A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Starting with chloroform,what reactions order and reagents are necessary to produce the following product and its enantiomer? ![Starting with chloroform,what reactions order and reagents are necessary to produce the following product and its enantiomer? A) [1] KOC(CH<sub>3</sub>) <sub>3</sub>,[2] trans-2-butene B) [1] KOC(CH<sub>3</sub>) <sub>3</sub>,[2] cis-2-butene C) [1] trans-2-butene,[2] KOC(CH<sub>3</sub>) <sub>3</sub> D) [1] cis-2-butene,[2] KOC(CH<sub>3</sub>) <sub>3</sub>](https://d2lvgg3v3hfg70.cloudfront.net/TB7662/11eac43c_f782_12a1_a9af_01bc7f46e148_TB7662_00.jpg)
A) [1] KOC(CH3) 3,[2] trans-2-butene
B) [1] KOC(CH3) 3,[2] cis-2-butene
C) [1] trans-2-butene,[2] KOC(CH3) 3
D) [1] cis-2-butene,[2] KOC(CH3) 3
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is the Grubbs catalyst synthetically important?
A) Because it only requires dilute concentrations of the reactants
B) Because it produces only stereospecific products
C) Because it produces only stereoselective products
D) Because it provides a synthetic pathway for ring-closing metathesis reactions
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the structure of the organic product Y formed in the following reaction sequence. 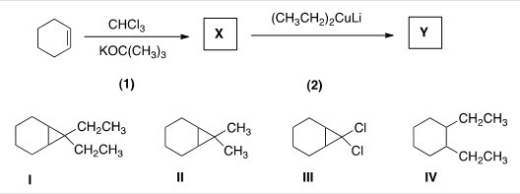
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product of the following reaction? 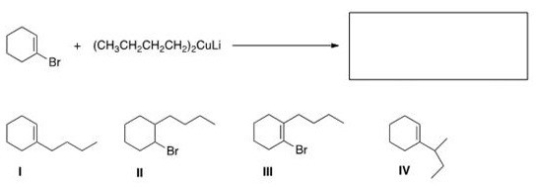
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
What is the major organic product of the following reaction? 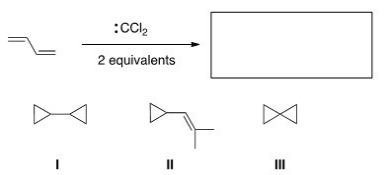
A) I
B) II
C) III
D) None of the choices is correct.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the starting material that would be used to form the following product in a ring-closing metathesis reaction utilizing a Grubbs catalyst? 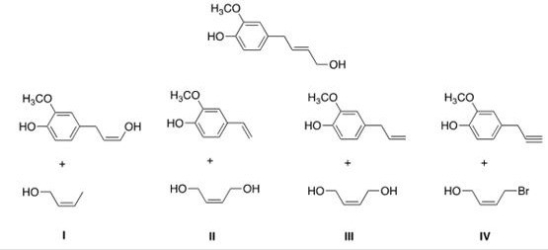
A) I
B) II
C) III
D) IV
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Organocuprate reagents (R2CuLi) react with several compounds.Which listed reaction is not correct?
A) Acid chlorides react with organocuprate reagents to form ketones.
B) Epoxides react with organocuprate reagents to form alcohols.
C) Alkyl halides react with organocuprate reagents to form coupling products containing a new carbon-carbon bond.
D) Carbon dioxide reacts with organocuprate reagents to form carboxylic acids.
F) C) and D)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
What is the major product of the following reaction? 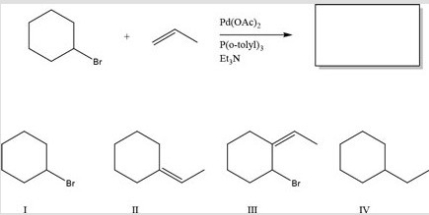
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What would be the starting material for the following product that was made via a Grubbs catalyst? 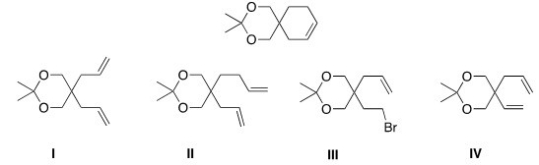
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Compound X can be synthesized via a ring-closing metathesis reaction when treated with a Grubbs' catalyst.What is a possible structure of the starting material for this reaction? 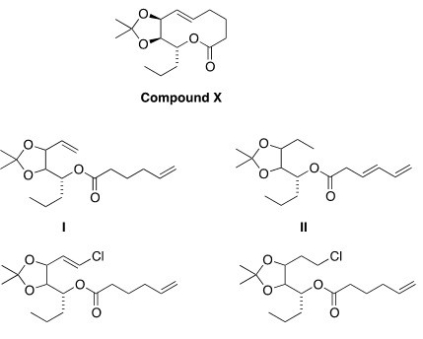
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Starting with chloroform,what reactions order and reagents are necessary to produce the following product? ![Starting with chloroform,what reactions order and reagents are necessary to produce the following product? A) [1] KOC(CH<sub>3</sub>) <sub>3</sub>,[2] trans-2-butene B) [1] KOC(CH<sub>3</sub>) <sub>3</sub>,[2] cis-2-butene C) [1] trans-2-butene,[2] KOC(CH<sub>3</sub>) <sub>3</sub> D) [1] cis-2-butene,[2] KOC(CH<sub>3</sub>) <sub>3</sub>](https://d2lvgg3v3hfg70.cloudfront.net/TB7662/11eac43c_f782_39b2_a9af_83be2ce6a9b3_TB7662_00.jpg)
A) [1] KOC(CH3) 3,[2] trans-2-butene
B) [1] KOC(CH3) 3,[2] cis-2-butene
C) [1] trans-2-butene,[2] KOC(CH3) 3
D) [1] cis-2-butene,[2] KOC(CH3) 3
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the structure of the major organic product that results from the following reaction. 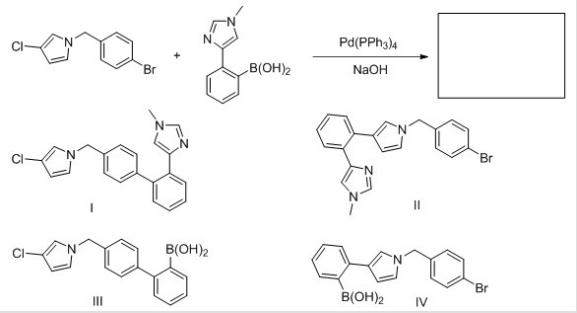
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is true about the use of the Grubbs catalyst?
A) The Grubbs catalyst is used in carbon-carbon coupling reactions.
B) The Grubbs catalyst is used in alkene metathesis.
C) The Grubbs catalyst is used in carbene formation.
D) The Grubbs catalyst is used with palladium as a co-catalyst.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the structure of the major organic product that results from the following reaction. 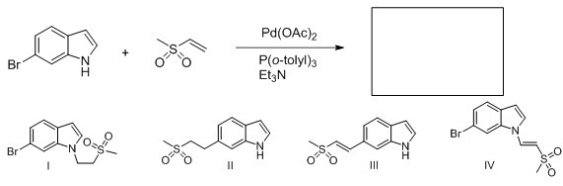
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
What is the missing reactant in the following reaction? 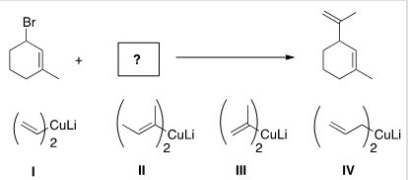
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following descriptions does not apply to methylene?
A) Methylene is sp2 hybridized.
B) Methylene is a neutral,reactive intermediate.
C) Methylene is a radical intermediate.
D) The formula of methylene is :CH2.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the products of the following reaction. 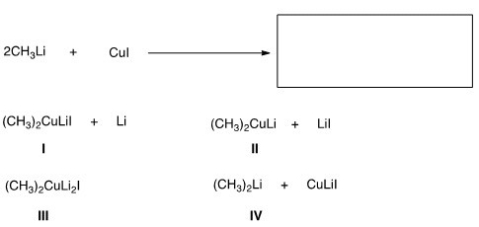
A) A
B) B
C) C
D) D
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Choose the statement below that is not true about the Suzuki reaction.
A) The product of the Suzuki reaction is completely stereospecific.
B) The Suzuki reaction involves both an organoborane reagent and an organopalladium catalyst.
C) The Suzuki reaction forms more highly substituted alkenes.
D) The Suzuki reaction involves an oxidative addition followed by a reductive elimination.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
As shown below,when cis-2-butene reacts with dichlorocarbene,only the cis-1,1-dichloro-2,3-dimethylcyclopropane is formed.What can we conclude about the nature of the reaction mechanism? 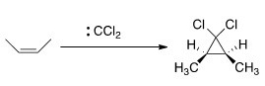
A) The mechanism is an SN1 mechanism.
B) The mechanism is a concerted.
C) The mechanism proceeds through a radical intermediate.
D) The mechanism is an E2 mechanism.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 47
Related Exams