A) IV > III > I > II
B) IV > II > I > III
C) II > I > IV > III
D) II > I > III > IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the labeled protons (Ha-Hd) in order of increasing acidity,starting with the least acidic. 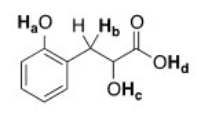
A) Ha < Hb < Hc < Hd
B) Hb < Hc < Ha < Hd
C) Hd < Ha < Hc < Hb
D) Hb < Hc < Hd < Ha
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What reagents are necessary to perform the following reaction? 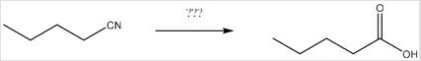
A) H2O,H+
B) LiAlH4,H2O
C) DIBAL-H,H2O
D) CH3MgBr,H2O
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is the C-O single bond of a carboxylic acid shorter than the C-O single bond of an alcohol?
A) The carbon in the alcohol is sp2 hybridized and has a higher percent s-character that lengthens the C-O bond in the alcohol.
B) The carbon in the carboxylic acid is sp3 hybridized and has a lower percent s-character that shortens the C-O bond in the carboxylic acid.
C) The carbon in the carboxylic acid is sp hybridized and has a higher percent s-character that shortens the C-O bond in the carboxylic acid.
D) The carbon in the carboxylic acid is sp2 hybridized and has a higher percent s-character that shortens the C-O bond in the carboxylic acid.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the correct IUPAC name of the following compound? 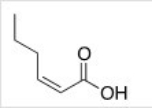
A) (E) -2-Hexenoic acid
B) (Z) -2-Hexenoic acid
C) (E) -4-Hexenoic acid
D) (Z) -4-Hexenoic acid
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What two groups make up the carboxylic acid group (RCOOH) ?
A) Carbon dioxide and hydrogen
B) Carbonyl and hydroxyl
C) Carbon monoxide and hydroxyl
D) Carbonyl oxide and hydrogen
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of increasing acidity,putting the least acidic first. 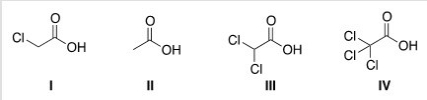
A) II < III < I < IV
B) II < I < III < IV
C) IV < III < I < II
D) IV < I < II < III
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the correct IUPAC name of the following compound? 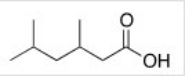
A) 4-Isopropyl-3-methylbutanoic acid
B) 2,4-Dimethylhexanoic acid
C) 3,5-Dimethylhexanoic acid
D) 3,5-Dimethy-1-hexanoic acid
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following compounds in order of increasing acidity,putting the least acidic first. 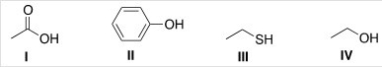
A) I < II < III < IV
B) IV < III < II < I
C) III < IV < II < I
D) IV < II < III < I
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Where do the two noteworthy peaks of carboxylic acids appear in 1HNMR spectra?
A) Between 10 and 12 ppm for the OH proton and 2-2.5 ppm for the protons on the a carbon to the carboxy group.
B) Between 6 and 9 ppm for the OH proton and 2-2.5 ppm for the protons on the a carbon to the carboxy group.
C) Between 10 and 12 ppm for the OH proton and 1-1.5 ppm for the protons on the a carbon to the carboxy group.
D) Between 6 and 9 ppm for the OH proton and 1-1.5 ppm for the protons on the a carbon to the carboxy group.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the structure of oxalic acid? 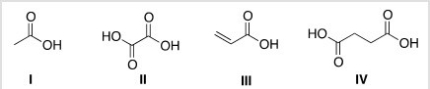
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
What is the correct IUPAC name of the following compound? 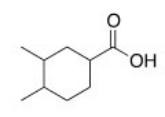
A) 3,4-Dimethylcyclohexanoic acid
B) 3,4-Dimethylcyclohexanecarboxylic acid
C) 4,5-Dimethylcyclohexanecarboxylic acid
D) 1,2-Dimethylcyclohexanecarboxylic acid
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
As applied to the chemistry of amino acids,what is the definition for the isoelectric point?
A) The pH at which the amino acid exists primarily in its acidic form.
B) The pH at which the amino acid exists primarily in its basic form.
C) The pH at which the amino acid exists as a mixture of isomers.
D) The pH at which the amino acid exists primarily in its neutral form.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the correct IUPAC name of the following compound? 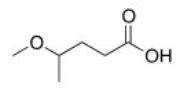
A) 2-Methoxypentanoic acid
B) 2-Methoxybutanoic acid
C) 4-Methoxybutanoic acid
D) 4-Methoxypentanoic acid
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the overall charge of the amino acid,alanine,at pH = 2?
A) + 1
B) - 1
C) No overall charge
D) + 2
F) All of the above
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Which of the following cannot be the starting material for the following reaction? 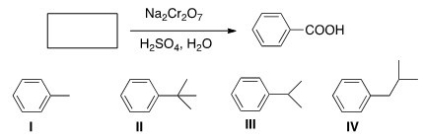
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following structures is the major contributor to the resonance hybrid of the phenoxide anion? 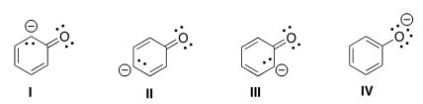
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What reagents are necessary to perform the following reaction? 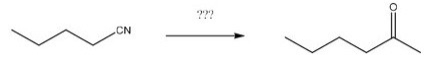
A) H2O,H+
B) LiAlH4,H2O
C) DIBAL-H,H2O
D) CH3MgBr,H2O
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What reagents are necessary to perform the following reaction? 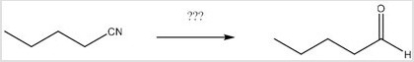
A) H2O,H+
B) LiAlH4,H2O
C) DIBAL-H,H2O
D) CH3MgBr,H2O
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of decreasing acidity,putting the most acidic first. 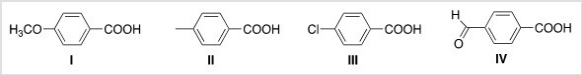
A) III > IV > I > II
B) IV > III > I > II
C) I > II > III > IV
D) IV > III > II > I
F) A) and C)
Correct Answer

verified
D
Correct Answer
verified
Showing 1 - 20 of 53
Related Exams