A) NaI
B) (1) NaNO2,HCl; (2) NaI
C) (1) NaNO2,HCl; (2) I2
D) I2
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why should the chirality of an ammonium salt with four different groups on the nitrogen atom not be ignored?
A) Because there is rapid interconversion between the two isomeric forms at room temperature
B) Because interconversion cannot occur between the two isomeric forms at room temperature
C) Because the compound would be a meso compound
D) Because the compound would be a racemic mixture
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the reagent(s) required for the following transformation. 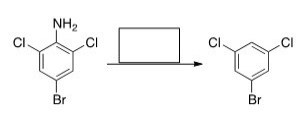
A) (1) NaNO2,HCl; (2) H2
B) (1) NaNO2,HCl; (2) H2O
C) (1) NaNO2,HCl; (2) H3PO4
D) (1) NaNO2,HCl; (2) H3PO2
F) A) and B)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
What is the major product of the following reaction? 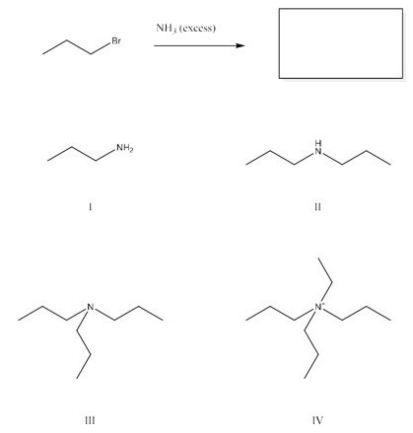
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name of the following compound? 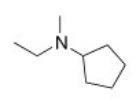
A) N-ethyl-N-methylcyclopentanamine
B) N-cyclopentyl-N-methylethanamine
C) N-methyl-N-ethylcyclopentylamine
D) N-ethyl-N-methylpentanamine
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the common name of the following compound? 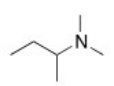
A) dimethylisobutylamine
B) butyldimethylamine
C) N,N-dimethylbutanamine
D) sec-butyldimethylamine
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the major product of the following reaction. 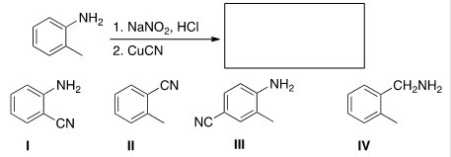
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Consider the following multistep synthesis. 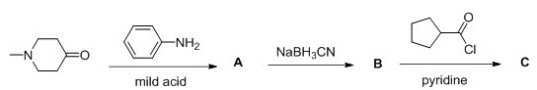 What is the structure of intermediate C?
What is the structure of intermediate C? 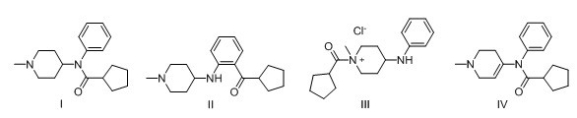
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the major product of the following reaction. 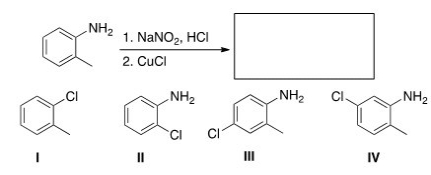
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What starting materials are required to synthesize the following azo compound? 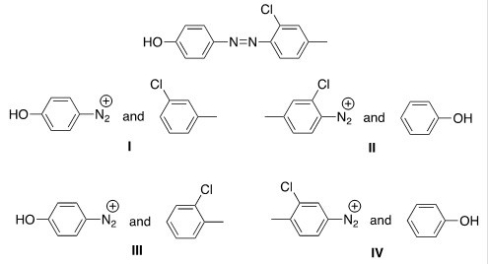
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of decreasing basicity,putting the most basic first. 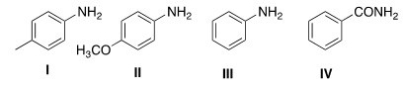
A) I > II > III > IV
B) I > III > II > IV
C) IV > III > I > II
D) II > I > III > IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the correct assignment of the names of the following aromatic amines? 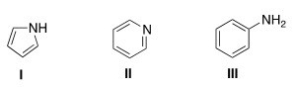
A) I = pyrrolidine; II = pyrimidine; III = aniline
B) I = pyrrole; II = pyrimidine; III = anisole
C) I = pyrrolidine; II = pyridine; III =aniline
D) I = pyrrole; II = pyridine; III = aniline
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following compounds in order of increasing boiling point,putting the compound with the least boiling point first. 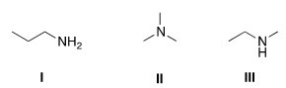
A) I < II < III
B) II < I < III
C) I < III < II
D) II < III < I
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is pyrrole much less basic than pyridine?
A) The lone pair of electrons in pyrrole is located on an sp2 orbital.
B) The lone pair of electrons in pyrrole is part of the aromatic p system.
C) The lone pair of electrons in pyrrole is not part of the aromatic p system.
D) The pKa of the conjugate acid of pyrrole is much greater than the conjugate acid of pyridine.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product obtained in the following reaction? 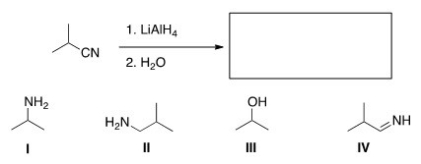
A) I
B) II
C) III
D) IV
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is the N-H bond of an imide especially acidic?
A) The conjugate base is stabilized by electron-donating inductive effect.
B) The conjugate base is stabilized by resonance.
C) The conjugate acid is stabilized by resonance.
D) The conjugate base is stabilized by intramolecular hydrogen bonding.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major product of the following reaction? 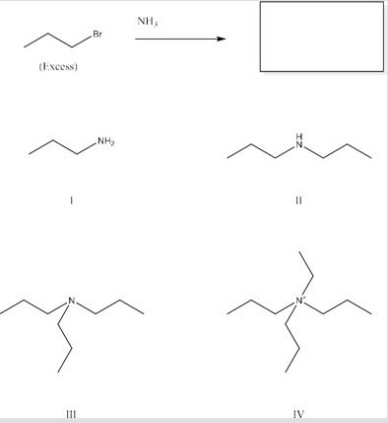
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the approximate bond angle of the substituents around a nitrogen atom in amines?
A) 90°
B) 109.5°
C) 120°
D) 180°
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name of the following compound? 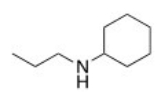
A) N-propylhexanamine
B) N-propylaniline
C) N-ethylcyclohexylamine
D) N-propylcyclohexanamine
F) All of the above
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
A compound with molecular formula C6H15N exhibits a singlet at d 0.9 (1H) ,a triplet at d 1.10 (3H) ,a singlet at d1.15 (9H) ,and a quartet at d 2.6 (2H) in its 1HNMR spectrum.Its IR spectrum shows one medium absorption band near 3400 cm-1.What is the structure of this compound? 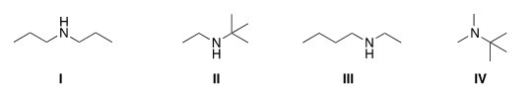
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 65
Related Exams