A) describe regions of space in which one is most likely to find an electron.
B) describe exact paths for electron motion.
C) give a description of the atomic structure which is essentially the same as the Bohr model.
D) allow scientists to calculate an exact volume for the hydrogen atom.
E) are in conflict with the Heisenberg Uncertainty Principle.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Platinum, which is widely used as a catalyst, has a work function 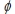 (the minimum energy needed to eject an electron from the metal surface) of 9.05 × 10-19 J. What is the longest wavelength of light which will cause electrons to be emitted?
(the minimum energy needed to eject an electron from the metal surface) of 9.05 × 10-19 J. What is the longest wavelength of light which will cause electrons to be emitted?
A) 2.196 × 10-7 m
B) 4.553 × 10-6 m
C) 5.654 × 102 m
D) 1.370 × 1015 m
E) > 106 m
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
In the Bohr model of the hydrogen atom, the electron moves in a circular path which Bohr referred to as an orbital.
B) False
Correct Answer

verified
False
Correct Answer
verified
Multiple Choice
The de Broglie equation predicts that the wavelength (in m) of a proton moving at 1000. m/s is
A) 3.96 × 10-10 m.
B) 3.96 × 10-7 m.
C) 2.52 × 106 m.
D) 2.52 × 109 m.
E) > 1010 m.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Infrared radiation from the sun has a wavelength of 6200 nm. Calculate the energy of one photon of that radiation.
A) 4.l × 10-39 J
B) 4.l × 10-30 J
C) 3.2 × 10-29 J
D) 3.2 × 10-26 J
E) between 10-20 and 10-19 J
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The AM station KBOR plays your favorite music from the 20's and 30's at 1290 kHz. Find the wavelength of these waves.
A) 4.30 × 10-2 m
B) 0.144 m
C) 6.94 m
D) 232 m
E) > 103 m
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The energy of a photon is directly proportional to the wavelength of the radiation.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Line spectra are characteristic of atoms in the gas phase.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A photon has an energy of 5.53 × 10-17 J. What is its frequency in s-1?
A) 3.66 × 10-50 s-1
B) 1.20 × 10-17 s-1
C) 3.59 × 10-9 s-1
D) 2.78 × 108 s-1
E) 8.35 × 1016 s-1
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
Continuous spectra are characteristic of heated solids.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Line spectra from all regions of the electromagnetic spectrum, including the Paschen series of infrared lines for hydrogen, are used by astronomers to identify elements present in the atmospheres of stars. Calculate the wavelength of the photon emitted when the hydrogen atom undergoes a transition from n = 5 to n = 3. (R = 1.096776 × 107 m-1)
A) 205.1 nm
B) 384.6 nm
C) 683.8 nm
D) 1282 nm
E) > 1500 nm
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In an atom, the square of an electron's wave function
A) becomes zero at the nucleus.
B) is smallest near the nucleus.
C) is largest near the nucleus.
D) may be zero at more than one point.
E) tends to infinity at large distances from the nucleus.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to the Bohr theory of the hydrogen atom, the minimum energy (in J) needed to ionize a hydrogen atom from the n = 2 state is
A) 2.18 × 10-18 J.
B) 1.64 × 10-18 J.
C) 5.45 × 10-19 J.
D) 3.03 × 10-19 J.
E) None of these choices are correct.
G) C) and D)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
A sprinter must average 24.0 mi/h to win a 100-m dash in 9.30 s. What is his wavelength at this speed if his mass is 84.5 kg?
A) 7.29 × 10-37 m
B) 3.26 × 10-37 m
C) 5.08 × 10-30 m
D) 1.34 × 10-30 m
E) None of these choices are correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the Rydberg equation to calculate the frequency of a photon absorbed when the hydrogen atom undergoes a transition from n1 = 2 to n2 = 4. (R = 1.096776 × 107 m-1)
A) 2.056 × 106 s-1
B) 2.742 × 106 s-1
C) 6.165 × 1014 s-1
D) 8.226 × 1014 s-1
E) > 1015 s-1
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following frequencies of electromagnetic radiation has the shortest wavelength?
A) 1 kilohertz
B) 1 terahertz
C) 1 dekahertz
D) 1 gigahertz
E) 1 megahertz
G) A) and C)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Which scientist first proposed that the electron in the hydrogen atom can have only certain energies?
A) Planck
B) Einstein
C) Bohr
D) Rydberg
E) Heisenberg
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a correct set of quantum numbers for an electron in a 3d orbital?
A) n = 3, l = 0, ml = -1
B) n = 3, l = 1, ml = +3
C) n = 3, l = 2, ml = 3
D) n = 3, l = 3, ml = +2
E) n = 3, l = 2, ml = -2
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The orientation in space of an atomic orbital is associated with
A) the principal quantum number (n) .
B) the angular momentum quantum number (l) .
C) the magnetic quantum number (ml) .
D) the spin quantum number (ms) .
E) None of these choices are correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Green light has a wavelength of 5200 Å. Calculate the energy of one photon of green light.
A) 3.4 × 10-40 J
B) 3.4 × 10-30 J
C) 3.8 × 10-29 J
D) 3.4 × 10-27 J
E) 3.8 × 10-19 J
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 69
Related Exams