A) H3PO4 phosphoric acid
B) HNO3 nitric acid
C) NaHCO3 sodium carbonate
D) H2CO3 carbonic acid
E) KOH potassium hydroxide
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Silicon, which makes up about 25% of Earth's crust by mass, is used widely in the modern electronics industry. It has three naturally occurring isotopes, 28Si, 29Si, and 30Si. Calculate the atomic mass of silicon. 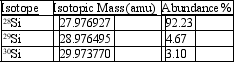
A) 29.2252 amu
B) 28.9757 amu
C) 28.7260 amu
D) 28.0855 amu
E) 27.9801 amu
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Diiodine pentaoxide is used as an oxidizing agent that converts carbon monoxide to carbon dioxide. What is its chemical formula?
A) I2O5
B) IO5
C) 2IO5
D) I5O2
E) (IO5) 2
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Who is credited with measuring the mass/charge ratio of the electron?
A) Dalton
B) Gay-Lussac
C) Thomson
D) Millikan
E) Rutherford
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name of Na2O?
A) disodium monoxide
B) sodium monoxide
C) sodium dioxide
D) sodium(I) oxide
E) sodium oxide
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Chlorine dioxide is a strong oxidizer that is used for bleaching flour and textiles and for purification of water. What is its formula?
A) (ClO) 2
B) Cl2O
C) Cl2O2
D) Cl2O4
E) ClO2
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name of the acid formed when HClO4 liquid is dissolved in water?
A) hydrochloric acid
B) perchloric acid
C) chloric acid
D) chlorous acid
E) hydrochlorate acid
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The compound, BaO, absorbs water and carbon dioxide readily and is used to dry gases and organic solvents. What is its name?
A) barium oxide
B) barium(II) oxide
C) barium monoxide
D) baric oxide
E) barium peroxide
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Copper (Cu) is a transition metal.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Silver chloride is used in photographic emulsions. What is its formula?
A) Ag2Cl3
B) Ag2Cl
C) AgCl3
D) AgCl2
E) AgCl
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
All neutral atoms of tin have 50 protons and 50 electrons.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Zinc acetate is used in preserving wood and in manufacturing glazes for porcelain. What is its formula?
A) ZnAc2
B) ZnCH3COO
C) Zn(CH3COO) 2
D) Zn2CH3COO
E) ZnCH3COCH3
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ammonium sulfate, (NH4) 2SO4, is a fertilizer widely used as a source of nitrogen. Calculate its molecular mass.
A) 63.07 amu
B) 114.10 amu
C) 118.13 amu
D) 128.11 amu
E) 132.13 amu
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Iron (III) chloride hexahydrate is used as a coagulant for sewage and industrial wastes. What is its formula?
A) Fe(Cl·6H2O) 3
B) Fe3Cl·6H2O
C) FeCl3(H2O) 6
D) Fe3Cl(H2O) 6
E) FeCl3·6H2O
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
The mass of a neutron is equal to the mass of a proton plus the mass of an electron.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ions occurs commonly?
A) P3+
B) Br7+
C) O6+
D) Ca2+
E) K-
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The compound, NaH2PO4, is present in many baking powders. What is its name?
A) sodium biphosphate
B) sodium hydrogen phosphate
C) sodium dihydrogen phosphate
D) sodium hydrophosphate
E) sodium dihydride phosphate
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Atoms of one element cannot be converted to another element by any known method.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The name for HF(g) is
A) hydrofluoric acid.
B) hydrogen(I) fluoride.
C) hydrogen fluoride.
D) hydrogen fluorine.
E) fluoric acid.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Atoms X, Y, Z, and R have the following nuclear compositions: Which two are isotopes? 
A) X & Y
B) X & R
C) Y & R
D) Z & R
E) X & Z
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 100
Related Exams