A) I and II
B) II and IV
C) II and III
D) III and IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the nucleophilic substitution reaction shown below?
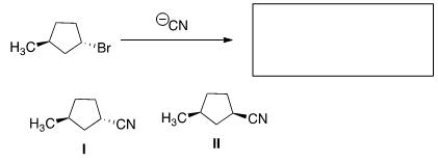
A) Only I
B) Only II
C) I and II
D) None
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction of 1-bromobutane with sodium hydroxide affords the substitution product 1-butanol.What would happen to the rate of the reaction if the concentration of both 1-bromobutane and sodium hydroxide were doubled?
A) The rate remains the same.
B) The rate decreases by a factor of 2.
C) The rate increases by a factor of 2.
D) The rate increases by a factor of 4.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about SN2 reactions is true?
A) The rate of reaction is dependent on just the substrate.
B) The fastest reaction will occur with a tertiary alkyl halide.
C) The mechanism is a two-step process.
D) Displacement occurs with inversion of configuration.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name of the following compound?
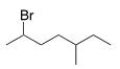
A) 2-Bromo-5-methyloctane
B) 2-Bromo-3-methylheptane
C) 2-Bromo-5-methylheptane
D) 6-Bromo-3-methylheptane
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction of what nucleophile and substrate is represented by the following transition state?
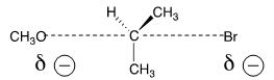
A) Methanol with 2-bromopropane
B) Methoxide with 2-bromopropane
C) Methoxide with 1-bromopropane
D) Methanol with 1-bromopropane
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name of the following compound?
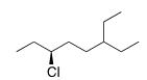
A) (R) -3-Chloro-6-ethyloctane
B) (S) -3-Chloro-6-ethyloctane
C) (S) -6-Chloro-3-ethyloctane
D) (R) -6-Chloro-3-ethyloctane
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following rate laws describes the kinetics of an SN1 reaction?
A) Rate = k[alkyl halide]
B) Rate = k[alkyl halide][nucleophile]
C) Rate = k[nucleophile]
D) Rate = k[solvent]
F) A) and D)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
What is the starting material in the reaction shown below?
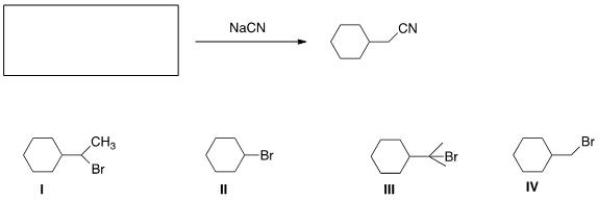
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of increasing SN1 reactivity?
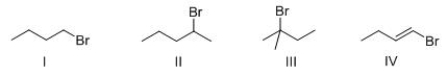
A) I > II > III > IV
B) IV > I > II > III
C) IV > III > II > I
D) III > II > I > IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction of tert-butyl bromide, (CH3) 3CBr, with ethanol affords the substitution product tert-butyl ethyl ether, (CH3) 3COCH2CH3, in acidic conditions.What would happen to the rate of the reaction if the concentration of ethanol was doubled?
A) The rate remains the same.
B) The rate decreases by a factor of 2.
C) The rate increases by a factor of 2.
D) The rate increases by a factor of 4.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name of the following compound?
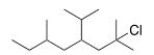
A) 2-Chloro-4-isopropyl-2,6-dimethyloctane
B) 2-Chloro-4-isopropyl-2,7-dimethylnonane
C) 2,6-Dimethyl-2-chloro-4-isopropyloctane
D) 7-Chloro-5-isopropyl-3,7-dimethyloctane
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following in order of decreasing nucleophilicity, putting the most nucleophilic first. 
A) II > III > I > IV
B) III > II > IV > I
C) II > III > IV > I
D) III > II > I > IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about an SN1 reaction mechanism is true?
A) The reaction involves two steps and occurs fastest with primary alkyl halides.
B) The reaction involves one step and occurs fastest with primary alkyl halides.
C) The reaction involves one step and occurs fastest with tertiary alkyl halides.
D) The reaction involves two steps and occurs fastest with tertiary alkyl halides.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following in order of increasing nucleophilicity, putting the least nucleophilic first. 
A) II < IV < I < III
B) IV < III < II < I
C) III < IV < I < II
D) II < I < IV < III
F) A) and B)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Which of the following alkyl halides would react the fastest with H2O in SN1 reaction? CH3CH2CH2CH2Br, (CH3) 2CHCH2Br, CH3CH2CH(CH3) Br, (CH3) 3CBr
A) CH3CH2CH2CH2Br
B) (CH3) 2CHCH2Br
C) CH3CH2CH(CH3) Br
D) (CH3) 3CBr
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following alkyl halides undergoes the fastest SN2 reaction with sodium hydroxide?
A) 1-Iodobutane
B) 1-Chlorobutane
C) 1-Fluorobutane
D) 1-Bromobutane
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is not true?
A) In polar protic solvents, nucleophilicity decreases down a column of the periodic table as the size of the anion increases.
B) Nucleophilicity is affected by the solvent used in a substitution reaction.
C) Polar protic solvents are capable of intermolecular hydrogen bonding.
D) Polar protic solvents solvate both cations and anions.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following alkyl halides is a primary alkyl halide?
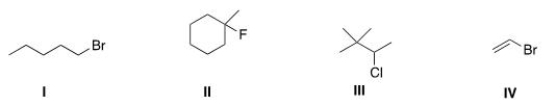
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Which of the following statements is true?
A) All good leaving groups are strong bases with weak conjugate acids.
B) Left-to-right across a row of the periodic table, leaving group ability decreases.
C) Down a column of the periodic table, leaving group ability decreases.
D) The conjugate bases of strong acids are good leaving groups.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 61
Related Exams