A) 4-Isopropyl-3-methylbutanoic acid
B) 2,4-Dimethylhexanoic acid
C) 3,5-Dimethylhexanoic acid
D) 3,5-Dimethy-1-hexanoic acid
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the presence of strong acids, which of the oxygen atoms on the carboxyl group is preferentially protonated and why?
A) Protonation occurs at the carbonyl oxygen because the resulting conjugate acid is stabilized by resonance.
B) Protonation occurs at the carbonyl oxygen because the resulting conjugate base is stabilized by the inductive effect.
C) Protonation occurs at the hydroxyl oxygen because the resulting conjugate acid is stabilized by resonance.
D) Protonation occurs at the hydroxyl oxygen because the resulting conjugate base is stabilized by the inductive effect.
F) A) and D)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
What is the common name of the following compound?
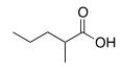
A) a-Methylbutyric acid
B) a-Propylpropionic acid
C) a-Methylvaleric acid
D) a-Methylcaproic acid
F) C) and D)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of increasing acidity, putting the least acidic first. 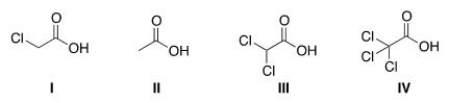
A) II < III < I < IV
B) II < I < III < IV
C) IV < III < I < II
D) IV < I < II < III
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the structure of oxalic acid?
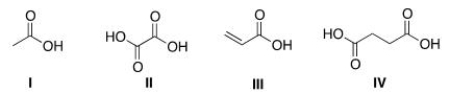
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What physical property and reaction type are used by extraction as useful techniques to separate and purify mixtures of compounds?
A) Physical property = solubility differences; reaction type = acid-base reaction.
B) Physical property = boiling point; reaction type = acid-base reaction.
C) Physical property = solubility differences; reaction type = oxidation-reduction.
D) Physical property = density; reaction type = oxidation-reduction.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the correct IUPAC name of the following compound?
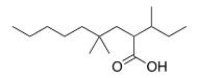
A) 2-sec-Butyl-4,4-dimethylnonanoic acid
B) 4,4-Dimethyl-2-isobutylnonanoic acid
C) 4,4-Dimethyl-2-sec-butylnonanoic acid
D) 2-Isobutyl-4,4-dimethylnonanoic acid
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following compounds in order of increasing acidity, putting the least acidic first. 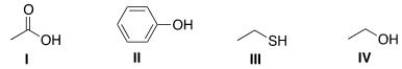
A) I < II < III < IV
B) IV < III < II < I
C) III < IV < II < I
D) IV < II < III < I
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
As applied to the chemistry of amino acids, what is the definition for the isoelectric point?
A) The pH at which the amino acid exists primarily in its acidic form.
B) The pH at which the amino acid exists primarily in its basic form.
C) The pH at which the amino acid exists as a mixture of isomers.
D) The pH at which the amino acid exists primarily in its neutral form.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of increasing acidity, putting the least acidic compound first. 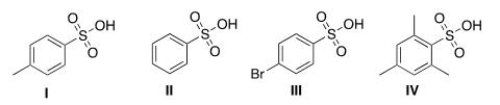
A) III < I < II < IV
B) IV < I < II < III
C) III < II < I < IV
D) II < IV < I < III
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the hybridization of the carbon atom in a carboxy group?
A) sp
B) sp2
C) sp3
D) p
F) A) and B)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of decreasing acidity, putting the most acidic first. 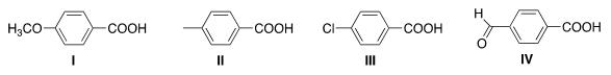
A) III > IV > I > II
B) IV > III > I > II
C) I > II > III > IV
D) IV > III > II > I
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the correct IUPAC name of the following compound?
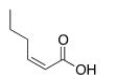
A) (E) -2-Hexenoic acid
B) (Z) -2-Hexenoic acid
C) (E) -4-Hexenoic acid
D) (Z) -4-Hexenoic acid
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the labeled protons (Ha-Hd) in order of increasing acidity, starting with the least acidic. 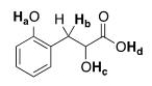
A) Ha < Hb < Hc < Hd
B) Hb < Hc < Ha < Hd
C) Hd < Ha < Hc < Hb
D) Hb < Hc < Hd < Ha
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following cannot be the starting material for the following reaction?
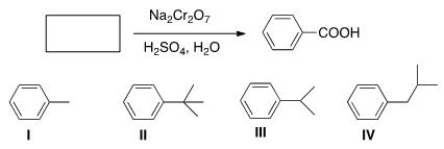
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following compounds in order of increasing water solubility, putting the least soluble compound first. 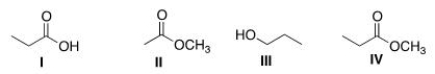
A) I < II < III < IV
B) II < I < III < IV
C) I < III < II < IV
D) IV < II < III < I
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is most basic?
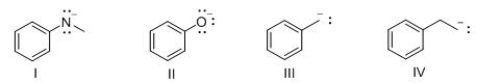
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following reagents can accomplish the transformation below?
![Which of the following reagents can accomplish the transformation below? A) PCC, CH<sub>2</sub>Cl<sub>2</sub> B) [1] LiAlH<sub>4</sub>, THF; [2] H<sub>2</sub>O C) [1] O<sub>3</sub>; [2] H<sub>2</sub>O D) K<sub>2</sub>Cr<sub>2</sub>O<sub>7</sub>, H<sub>2</sub>SO<sub>4</sub>, H<sub>2</sub>O](https://d2lvgg3v3hfg70.cloudfront.net/TB5872/11eb4ad7_6c17_917d_99b2_a7949461a64e_TB5872_00.jpg)
A) PCC, CH2Cl2
B) [1] LiAlH4, THF; [2] H2O
C) [1] O3; [2] H2O
D) K2Cr2O7, H2SO4, H2O
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the overall charge of the amino acid, alanine, at pH = 7?
A) + 1
B) - 1
C) No overall charge
D) + 2
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following compounds in order of increasing boiling point, putting the compound with the lowest boiling point first. 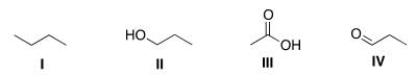
A) I < II < III < IV
B) I < IV < II < III
C) III < II < IV < I
D) II < IV < I < III
F) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 36
Related Exams