A) 658 K
B) 683 K
C) 955 K
D) 1047 K
E) 1229 K
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
For a given reaction, a change in the pressure may result in a change in the sign of ΔG.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
In some spontaneous processes, the entropy of the surroundings decreases.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following pairs has the member with the greater molar entropy listed first? All systems are at 25°C.
A) CO( g) , CO 2( g)
B) NaCl( s) , NaCl( aq)
C) H 2S( g) , H 2S( aq)
D) Li( s) , Pb( s)
E) H 2( g) , H 2O( g)
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is always true for an exothermic process?
A) q sys > 0, Δ S surr < 0
B) q sys < 0, Δ S surr > 0
C) q sys < 0, Δ S surr < 0
D) q sys > 0, Δ S surr > 0
E) w < 0
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a process with ΔS < 0, which one of the following statements is correct?
A) The process will definitely be spontaneous if ΔH < 0.
B) The process will be definitely be spontaneous if Δ H < TΔ S.
C) The process can never be spontaneous.
D) The process will definitely be spontaneous, regardless of Δ H.
E) The process will definitely be spontaneous if Δ S surr > 0.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Given: H2O(l) → H2O(g) ΔH° = 40.7 kJ at 373K What is the entropy change in the system (ΔS) when one mole of water vaporizes at 100°C and a pressure of one atmosphere?
A) 407 J/K
B) −407 J/K
C) 109 J/K
D) −109 J/K
E) J/K
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In order for a process to be spontaneous,
A) Δ H must be less than zero.
B) Δ S must be greater than zero.
C) Δ G must be greater than zero.
D) it should be rapid.
E) Δ S sys + Δ S surr must be greater than zero.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate ΔG° for the reaction of ammonia with fluorine. 2NH3(g) + 5F2(g) → N2F4(g) + 6HF(g)
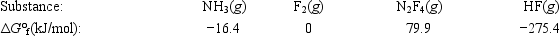
A) 179.1 kJ
B) −179.1 kJ
C) 1539.7 kJ
D) −1539.7 kJ
E) None of these choices are correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the thermodynamic data at 298 K below to determine the Ksp for barium carbonate, BaCO3 at this temperature. 
A) 5.86
B) 6.30 × 10 8
C) 1.59 × 10 −9
D) 5.47 × 10 −21
E) 2.18 × 10 −27
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the given data at 298 K to calculate ΔG° for the reaction 2Cl2(g) + SO2(g) → SOCl2(g) + Cl2O(g)
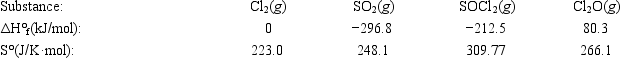
A) 129.3 kJ
B) 133.6 kJ
C) 196.0 kJ
D) 199.8 kJ
E) 229.6 kJ
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The entropy of one mole of oxygen gas in a 0.5-L container is less than it would be in a 22.4-L container at the same temperature.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The temperature at which the following process reaches equilibrium at 1.0 atm is the normal boiling point of hydrogen peroxide. H2O2(l) ⇄ H2O2(g)
Use the following thermodynamic information at 298 K to determine this temperature.
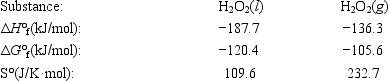
A) 120°C
B) 144°C
C) 196°C
D) 418°C
E) 585°C
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When a sky diver free-falls through the air, the process is
A) non-spontaneous because he is accelerating due to the force applied by gravity.
B) non-spontaneous because he is losing potential energy.
C) non-spontaneous, if he had planned the jump for two weeks.
D) spontaneous.
E) in equilibrium.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
In a spontaneous process, the entropy of the system always increases.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A certain process has ΔSuniv > 0 at 25°C. What does one know about the process?
A) It is exothermic.
B) It is endothermic.
C) It is spontaneous at 25°C.
D) It will move rapidly toward equilibrium.
E) None of these choices are correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In order for a process to be spontaneous,
A) the entropy of the system must increase.
B) the entropy of the surroundings must increase.
C) the entropy of the universe must decrease.
D) the entropy of the surroundings must decrease.
E) the entropy change of the surroundings plus the entropy change of the system must be positive.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct statement of a law of thermodynamics.
A) The enthalpy of the universe is constantly increasing.
B) The entropy of the universe is constant.
C) The entropy of an element in its standard state at 25°C is zero.
D) Every spontaneous process is accompanied by an increase in total entropy.
E) Every spontaneous process is accompanied by a decrease in the enthalpy of the system.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which relationship best describes ΔS° for the following reaction? CO(g) + H2O(g) → CO2(g) + H2(g)
A) Δ S° = Δ H°
B) Δ S° = Δ H°/ T
C) Δ S° > 0
D) Δ S° < 0
E) Δ S° ≈ 0
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Elemental boron can be formed by reaction of boron trichloride with hydrogen. BCl3(g) + 1.5H2(g) → B(s) + 3HCl(g)
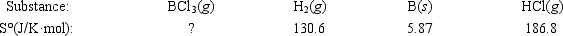 If ΔS° = 80.3 J/K for the reaction above, what is S° for BCl3(g) ?
If ΔS° = 80.3 J/K for the reaction above, what is S° for BCl3(g) ?
A) −18.2 J/K·mol
B) 18.2 J/K·mol
C) 290.1 J/K·mol
D) 355.4 J/K.mol
E) 450.6 J/K·mol
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 85
Related Exams