A) 90°
B) 109°
C) 120°
D) 180°
E) between 110 and 120°
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
According to VSEPR theory, a molecule with the general formula AX3E2 (where E represents a lone pair on A) will be trigonal planar.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory, a molecule with the general formula AX3E2 will have a __________ molecular shape.
A) trigonal pyramidal
B) trigonal bipyramidal
C) trigonal planar
D) T-shaped
E) see-saw
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The formal charge on Cl in the structure shown for the perchlorate ion is 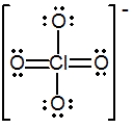
A) −2
B) −1
C) 0
D) +1
E) +2
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which one of the following is the best Lewis structure a resonance structure? (central atoms are bold)
A) CO 2
B) ClO 3 −
C) COCl 2
D) NO 2 +
E) HCN
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of XeO2F2 as predicted by the VSEPR theory? 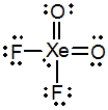
A) Square planar
B) Tetrahedral
C) Square pyramidal
D) See-saw
E) Octahedral
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the COCl2 molecule, carbon is the central atom. Based on the best Lewis structure for COCl2, what is the formal charge on carbon?
A) 0
B) +1
C) −1
D) +2
E) −2
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of SiF62− as predicted by the VSEPR theory? 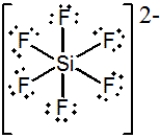
A) Trigonal bipyramidal
B) Hexagonal
C) Tetrahedral
D) See-saw
E) Octahedral
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
In formaldehyde, CH2O, both the formal charge and the oxidation number of carbon are zero.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In neutral molecules, how many bonds are commonly formed by nitrogen? And how many by oxygen?
A) 5 and 6
B) 4 and 2
C) 3 and 6
D) 4 and 6
E) 3 and 2
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many faces and how many vertexes (corners) , respectively, are there in a trigonal bipyramid?
A) 4 and 4
B) 5 and 5
C) 5 and 6
D) 6 and 5
E) 6 and 8
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of ClF4− as predicted by the VSEPR theory?
A) Square pyramidal
B) Square planar
C) See-saw
D) Octahedral
E) Tetrahedral
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which one of the following molecules are all the bonds single?
A) O 3
B) POCl 3
C) CO
D) COCl 2
E) N 2H 4
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use VSEPR theory to decide which one of the following molecules and ions will have a trigonal pyramidal geometry. (The central atom is always first in the formula.)
A) PCl 3
B) BF 3
C) SO 3
D) BrF 3
E) CO 3 2−
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the ideal bond angles in FNO using the molecular shape given by the VSEPR theory.
A) 90°
B) 109°
C) 120°
D) 180°
E) between 120 and 180°
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Bond angles of 180° only occur around atoms which display linear molecular geometry.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct Lewis structure for TeBr2.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use VSEPR theory to predict the electron group arrangement around iodine, the central atom in the ion IF2−.
A) Octahedral
B) Trigonal bipyramidal
C) Tetrahedral
D) Trigonal planar
E) Bent
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following atoms can expand its valence shell when bonding?
A) N
B) C
C) O
D) P
E) Al
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of ClF2− as predicted by the VSEPR theory?
A) Linear
B) Bent
C) See-saw
D) T-shaped
E) L-shaped
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 98
Related Exams