A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) None of these choices are correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory, a molecule with the general formula AX2 will have a __________ molecular shape.
A) linear
B) bent
C) trigonal planar
D) tetrahedral
E) triangular
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of N2O as predicted by the VSEPR theory? 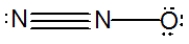
A) Trigonal pyramidal
B) Trigonal planar
C) Angular
D) Bent
E) Linear
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The best Lewis structure for sulfuric acid has zero formal charges, sulfur as the central atom, and no bonds between S and H. How many single and double bonds, respectively, are there in this Lewis structure?
A) 2 single, 4 double
B) 4 single, 2 double
C) 4 single, no double
D) 6 single, no double
E) 5 single, 1 double
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the actual bond angle in SeCl2 using the VSEPR theory.
A) more than 120°
B) between 109° and 120°
C) between 90° and 109°
D) exactly 90°
E) less than 90°
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which one of the following species is the best Lewis structure a resonance structure?
A) NH 3
B) CO 2
C) SF 6
D) O 2
E) CO 3 2−
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory, a molecule with the general formula AX2E3 will have a __________ molecular shape.
A) bent
B) linear
C) trigonal planar
D) T-shaped
E) trigonal pyramidal
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Thionyl chloride is used as an oxidizing and chlorinating agent in organic chemistry. Select the best Lewis structure for SOCl2.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) None of these choices are correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the hemoglobin molecule that carries oxygen molecules in blood, iron has 6 single bonds to surrounding atoms. What is the most likely geometry of these bonds to iron?
A) Planar
B) Linear
C) Tetrahedral
D) Octahedral
E) Trigonal pyramidal
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following Lewis structures is definitely incorrect?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory, a molecule with the general formula AX5E will have a __________ molecular shape.
A) tetrahedral
B) trigonal bipyramidal
C) square pyramidal
D) octahedral
E) see-saw
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In carbon disulfide, how many lone pairs of electrons are on each sulfur atom?
A) 0
B) 1
C) 2
D) 3
E) 4
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory, a molecule with the general formula AX3E will have a __________ molecular shape.
A) bent
B) trigonal planar
C) trigonal pyramidal
D) tetrahedral
E) triangular
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which one of the following is the best Lewis structure a resonance structure?
A) SO 3
B) BF 3
C) I 3 −
D) SCO (C = central atom)
E) SO 3 2−
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following has no net dipole moment?
A) N 2O
B) NF 3
C) H 2Se
D) TeO 3
E) CH 3Cl
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct Lewis structure for nitrogen trifluoride, NF3.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the Lewis structure in which formal charges are minimized for the periodate anion, IO4−.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the smallest actual bond angle in BrF3 using the VSEPR theory.
A) more than 120°
B) exactly 120°
C) between 109° and 120°
D) between 90° and 109°
E) less than 90°
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 98 of 98
Related Exams