A) Kc = K1 + K2.
B) Kc = K1/K2.
C) Kc = K1 - K2.
D) Kc = (K1) (K2) .
E) Kc = K2/K1.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
For the reaction of A + B  C + D + heat, the reverse reaction is exothermic.
C + D + heat, the reverse reaction is exothermic.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Suppose 25.0 g of HI(g) is injected into a 4.00-L reaction vessel that contains 20.0 g of I2(g) . When the system comes to equilibrium at 400.ºC, what will be the total pressure inside the reaction vessel? (R = 0.08206 L • atm/K • mol) 2HI(g)  H2(g) + I2(g) , Kc = 0.0156 at 400.ºC
H2(g) + I2(g) , Kc = 0.0156 at 400.ºC
A) 2.70 atm
B) 13.0 atm
C) 2.43 atm
D) 0.815 atm
E) 3.79 atm
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which represents the correct relationship between the Gibbs free energy and the equilibrium constant?
A) ΔG = -RT lnK
B) ΔG = RT lnK
C) ΔGo = -RT lnK
D) ΔGo = -RT lnQ
E) ΔGo = RT lnQ
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Nitric oxide and bromine were allowed to react in a sealed container. When equilibrium was reached, the following partial pressures of three gases were measured: NO: 0.526 atm; Br2 : 1.59 atm; NOBr: 7.68 atm. Calculate KP for the reaction. 2NO(g) + Br2(g)  2NOBr(g)
2NOBr(g)
A) 7.45 × 10-3
B) 0.109
C) 9.18
D) 91.8
E) 134
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The equilibrium constant KP at 427°C for the reaction N2(g) + 3H2(g)  2NH3(g) is 9.4 × 10-5. What is ΔG° for the reaction under these conditions? (R = 8.314 J/K • mol)
2NH3(g) is 9.4 × 10-5. What is ΔG° for the reaction under these conditions? (R = 8.314 J/K • mol)
A) -33 kJ/mol
B) -54 kJ/mol
C) 54 kJ/mol
D) 33 kJ/mol
E) 1.3 J/mol
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the equilibrium constant at 25°C for the reaction of methane with water to form carbon dioxide and hydrogen. The data below are values at 25°C.(R = 8.314 J/K • mol) CH4(g) + 2H2O(g)  CO2(g) + 4H2(g)
CO2(g) + 4H2(g) 
A) 8.2 × 1019
B) 0.96
C) 0.58
D) 1.2 × 10-20
E) 1.4 × 10-46
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate KP for the reaction 2NOCl(g)  2NO(g) + Cl2(g) at 400.°C if Kc at 400.°C for this reaction is 2.1 × 10-2. (R = 0.08206 L • atm/K • mol)
2NO(g) + Cl2(g) at 400.°C if Kc at 400.°C for this reaction is 2.1 × 10-2. (R = 0.08206 L • atm/K • mol)
A) 6.4 × 10-3
B) 1.7 × 10-3
C) 0.69
D) 1.2
E) 3.8 × 10-4
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the endothermic reaction A2 (g)  2A(g) , a snapshot of an equilibrium mixture of A(g) and A2 (g) at low temperature may look as follows. (Each circle represents 1.0 mol of A atoms, and the volume of the box is 1.0 L.)
2A(g) , a snapshot of an equilibrium mixture of A(g) and A2 (g) at low temperature may look as follows. (Each circle represents 1.0 mol of A atoms, and the volume of the box is 1.0 L.) 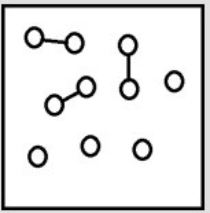 If the temperature is raised, what might the new equilibrium system look like?
If the temperature is raised, what might the new equilibrium system look like?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The standard free energy of formation of gaseous hydrogen iodide is 1.30 kJ/mol at 25°C. What is KP for the reaction H2(g) + I2(g)  2HI(g) at this temperature?
2HI(g) at this temperature?
A) 0.35
B) 0.59
C) 1.0
D) 1.7
E) 2.9
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
A(n) ________ means that the products of the reaction can react to form reactants.
Correct Answer

verified
Correct Answer
verified
True/False
A change in a concentration will not change the position of equilibrium.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the following reaction at equilibrium, which choice gives a change that will shift the position of equilibrium to favor formation of more products? 2NOBr(g)  2NO(g) + Br2(g) , ΔHºrxn = 30 kJ/mol
2NO(g) + Br2(g) , ΔHºrxn = 30 kJ/mol
A) Increase the total pressure by decreasing the volume
B) Add more NO
C) Remove Br2
D) Lower the temperature
E) Remove NOBr selectively
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Compounds A, B, and C react according to the following equation. 3A(g) + 2B(g) ![Compounds A, B, and C react according to the following equation. 3A(g) + 2B(g) 2C(g) At 100°C a mixture of these gases at equilibrium showed that [A] = 0.855 M, [B] = 1.23 M, and [C] = 1.75 M. What is the value of K<sub>c</sub> for this reaction? A) 0.309 B) 0.601 C) 1.66 D) 2.25 E) 3.24](https://d2lvgg3v3hfg70.cloudfront.net/TB8482/11eb6c5c_3a80_750a_8d9d_3fcafa62e0f3_TB8482_11.jpg) 2C(g)
At 100°C a mixture of these gases at equilibrium showed that [A] = 0.855 M, [B] = 1.23 M, and [C] = 1.75 M. What is the value of Kc for this reaction?
2C(g)
At 100°C a mixture of these gases at equilibrium showed that [A] = 0.855 M, [B] = 1.23 M, and [C] = 1.75 M. What is the value of Kc for this reaction?
A) 0.309
B) 0.601
C) 1.66
D) 2.25
E) 3.24
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which of these gas-phase equilibria is the yield of products increased by increasing the total pressure on the reaction mixture?
A) CO(g) + H2O(g) ![]() CO2(g) + H2(g)
CO2(g) + H2(g)
B) 2NO(g) + Cl2(g) ![]() 2NOCl(g)
2NOCl(g)
C) 2SO3(g) ![]() 2SO2(g) + O2(g)
2SO2(g) + O2(g)
D) PCl5(g) ![]() PCl3(g) + Cl2(g)
PCl3(g) + Cl2(g)
E) 2H2O2 (g) ![]() 2H2O(g) + O2(g)
2H2O(g) + O2(g)
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following reaction is at equilibrium in a sealed container. N2(g) + 3H2(g)  2NH3(g) ; ΔH°rxn < 0
Which, if any, of the following actions will increase the value of the equilibrium constant, Kc?
2NH3(g) ; ΔH°rxn < 0
Which, if any, of the following actions will increase the value of the equilibrium constant, Kc?
A) Adding more NH3
B) Adding more N2
C) Increasing the pressure
D) Lowering the temperature
E) Adding a catalyst
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is defined as a fraction with product concentrations in the numerator and reactant concentrations in the denominator and with each concentration raised to a power equal to the corresponding stoichiometric coefficient in the balanced chemical equation?
A) Reversibility expression
B) Reaction expression
C) Equilibrium expression
D) Reaction quotient
E) Mass action
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hydrogen peroxide may decompose to form water and oxygen gas according to the following reaction. 2H2O2(g)  2H2O(g) + O2(g)
In a particular experiment, 1.75 moles of H2O2 were placed in a 2.5-L reaction chamber at 307ºC. After equilibrium was reached, 1.20 moles of H2O2 remained. What is Kc for the reaction?
2H2O(g) + O2(g)
In a particular experiment, 1.75 moles of H2O2 were placed in a 2.5-L reaction chamber at 307ºC. After equilibrium was reached, 1.20 moles of H2O2 remained. What is Kc for the reaction?
A) 2.0 × 10-4
B) 2.3 × 10-2
C) 2.4 × 10-3
D) 5.5 × 10-3
E) 3.9 × 10-4
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is incorrect regarding equilibrium?
A) Chemical equilibrium is a reversible process with no net change in concentrations of the products and reactants.
B) Physical equilibrium cannot exist between phases.
C) A chemical equilibrium with all reactants and products in the same phase is homogeneous.
D) None of these choices.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the common allotropes of carbon (graphite and diamond) , C(gr)  C(dia) with equilibrium constant K = 0.32. The molar volumes of graphite and diamond are, respectively, 5.30 cm3/mol and 3.42 cm3/mol; ΔHf of diamond is 1.90 kJ/mol. These data suggest that the formation of diamond is favored at
C(dia) with equilibrium constant K = 0.32. The molar volumes of graphite and diamond are, respectively, 5.30 cm3/mol and 3.42 cm3/mol; ΔHf of diamond is 1.90 kJ/mol. These data suggest that the formation of diamond is favored at
A) low temperatures and low pressures.
B) high temperatures and low pressures.
C) low temperatures and high pressures.
D) high temperatures and high pressures.
E) None of these choices.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 121 - 140 of 146
Related Exams