A) 15 s
B) 30 s
C) 36 s
D) 45 s
E) 60 s
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following is an Arrhenius plot of a first-order reaction. The rate constant is measured in units of s-1. 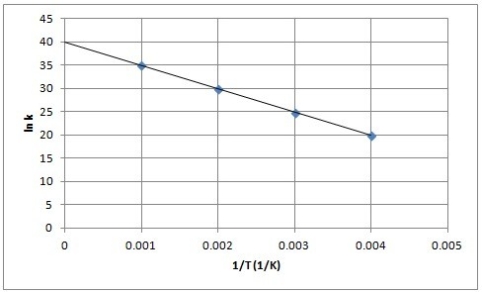 Based on this Arrhenius plot, what is the Arrhenius frequency factor (A) of the reaction? (R = 8.314 J/K mol)
Based on this Arrhenius plot, what is the Arrhenius frequency factor (A) of the reaction? (R = 8.314 J/K mol)
A) 5.0 s-1
B) 40 s-1
C) 50 s-1
D) 5.0 × 103 s-1
E) 2.4 × 1017 s-1
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the reaction 2NOBr(g) → 2NO(g) + Br2(g) . The initial rate of the reaction was measured for three different concentrations of NOBr, given below.  Based on the initial rate data above, what is the value of the rate constant?
Based on the initial rate data above, what is the value of the rate constant?
A) 0.0360 L • mol-1 • s-1
B) 0.800 L • mol-1 • s-1
C) 1.25 L • mol-1 • s-1
D) 27.8 L • mol-1 • s-1
E) 0.0360 s-1
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The activation energy for the following first-order reaction is 102 kJ/mol. N2O5(g) → 2NO2(g) + (1/2) O2(g) The value of the rate constant (k) is 1.35 × 10-4 s-1 at 35°C. What is the value of k at 0°C? (R = 8.314 J/mol• K)
A) 8.2 × 10-7 s-1
B) 1.9 × 10-5 s-1
C) 4.2 × 10-5 s-1
D) 2.2 × 10-2 s-1
E) 1.2 × 106s-1
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
The ________ is the rate for a specific instant in time.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the integrated rate law for a zeroth-order reaction?
A) rate = k[A]t
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction 2NO2(g) → 2NO(g) + O2(g) is suspected to be second order in NO2. Which of the following kinetic plots would be the most useful to confirm whether or not the reaction is second order?
A) A plot of [NO2]-1 vs. t
B) A plot of ln [NO2] vs. t
C) A plot of [NO2] vs. t
D) A plot of ln [NO2]-1 vs. t
E) A plot of [NO2]2 vs. t
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What statement below best describes the graph representing the integrated first-order rate law?
A) A plot of [A]t vs. t yields a straight line with a negative slope equal to -k.
B) A plot of 1/[A]t vs. t yields a straight line with a negative slope equal to k.
C) A plot of ln[A]t vs. t yields a straight line with a positive slope equal to -k.
D) A plot of ln[A]t vs. t yields a straight line with a negative slope equal to -k.
E) A plot of 1/[A]t vs. t yields a straight line with a positive slope equal to k.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ammonium cyanate (NH4CNO) reacts to form urea (NH2CONH2) . At 65°C the rate constant, k, is 3.60 L • mol-1 • s-1. What is the rate law for this reaction?
A) Rate = (3.60 L • mol-1 • s-1) [NH4CNO]
B) Rate = (3.60 L • mol-1 • s-1) [NH4CNO]2
C) Rate = (0.28 mol • L-1 • s-1) [NH4CNO]
D) Rate = (0.28 mol • L-1 • s-1) [NH4CNO]2
E) Rate = (3.60 L • mol-1 • s-1) [NH2CONH2]-1
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the half-life for a second-order reaction?
A) t1/2 = k
B) t1/2 = 1/k[A]o
C) t1/2 = 0.693/k[A]o
D) t1/2 = [A]o/2k
E) t1/2 = 0.693/k
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction A + 2B → Products has the rate law, rate = k[A][B]3. If the concentration of B is doubled while that of A is unchanged, by what factor will the rate of reaction increase?
A) 2
B) 4
C) 6
D) 8
E) 9
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the hypothetical reaction A + 3B → 2C, the rate should be expressed as
A) rate = Δ[A]/Δt.
B) rate = -Δ[C]/Δt.
C) rate = -3(Δ[B]/Δt) .
D) rate = (1/2) (Δ[C]/Δt) .
E) rate = (1/3) (Δ[B]/Δt) .
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the reaction X + Y → Z, the reaction rate is found to depend only upon the concentration of X. A plot of 1/X versus time gives a straight line. ![For the reaction X + Y → Z, the reaction rate is found to depend only upon the concentration of X. A plot of 1/X versus time gives a straight line. What is the rate law for this reaction? A) rate = k [X] B) rate = k [X]<sup>2</sup> C) rate = k [X][Y] D) rate = k [X]<sup>2</sup>[Y] E) rate = k [X]<sup>2</sup>/[Y]](https://d2lvgg3v3hfg70.cloudfront.net/TB8482/11eb6c5c_3a9a_8c59_8d9d_050991b4d202_TB8482_00.jpg) What is the rate law for this reaction?
What is the rate law for this reaction?
A) rate = k [X]
B) rate = k [X]2
C) rate = k [X][Y]
D) rate = k [X]2[Y]
E) rate = k [X]2/[Y]
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The rate law for the reaction 3A → C is rate = 4.36 × 10-2 L• mol-1 • h-1[A]2. What is the half-life for the reaction if the initial concentration of A is 0.250 M?
A) 0.0109 h
B) 0.0629 h
C) 15.9 h
D) 23.9 h
E) 91.7 h
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
According to the ________ ________ of chemical kinetics, the reaction rate is directly proportional to the number of molecular collisions per second.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Butadiene, C4H6 (used to make synthetic rubber and latex paints) , dimerizes to C8H12 with a rate law of rate = 0.014 L/mol• s [C4H6]2. What will be the concentration of C4H6 after 3.0 hours if the initial concentration is 0.025 M?
A) 0.0052 M
B) 0.024 M
C) 43 M
D) 190 M
E) 0.0000 M
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A certain first-order reaction A → B is 25% complete in 42 min at 25°C. What is its rate constant?
A) 6.8 × 10-3 min-1
B) 8.3 × 10-3 min-1
C) 3.3 × 10-2 min-1
D) -3.3 × 10-2 min-1
E) 11 min-1
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sucrose decomposes to fructose and glucose in acid solution. When ln [sucrose] is plotted vs. time, a straight line with slope of -0.208 h-1 results. What is the rate law for the reaction?
A) Rate = (0.208 h-1) [sucrose]2
B) Rate = (0.208 h-1) [sucrose]
C) Rate = (0.0433 h) [sucrose]2
D) Rate = (0.0433 h) [sucrose]
E) Rate = (0.208 mol L-1 • h-1) [sucrose]0
G) D) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The units of the rate of reaction depend on the order of the reaction.
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
A(n) ________ is the name given to a collision that does result in a reaction.
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 132
Related Exams