A) (CH3) 3N
B) CH3-O-CH3
C) CH3CH2-OH
D) CH3CH2-F
E) HI
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following lacks a regular three-dimensional arrangement of atoms?
A) crystalline solid
B) ionic crystal
C) solid
D) amorphous solid
E) molecular solid
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the energy in kJ/mol required to melt 1 mole of a solid?
A) molar heat of freezing
B) molar heat of fission
C) molar heat of vaporization
D) molar heat of fusion
E) molar heat of condensation
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The energy of a hydrogen bond is greater than that of a typical covalent bond.
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
Identify the dominant (strongest) type of intermolecular force present in NH3(l).
Correct Answer

verified
Correct Answer
verified
Short Answer
Give an example of an ionic crystal and a metallic crystal which contain an element common to each.
Correct Answer

verified
NaCl = ion...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which statement is true about phase diagrams?
A) The triple point pressure is always found below 1 atmosphere.
B) The slope of the liquid-solid phase boundary line is typically negative.
C) The triple point temperature is the lowest temperature where a liquid can exist.
D) The critical temperature is lower than the triple point temperature.
E) The critical temperature is lower than the normal boiling point.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of atoms in a body-centered cubic unit cell is
A) 1.
B) 2.
C) 3.
D) 4.
E) 8.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of atoms in a face-centered cubic unit cell is
A) 1.
B) 2.
C) 3.
D) 4.
E) 8.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The location that indicates conditions under which two phases can exist in equilibrium is called the
A) triple point.
B) critical point.
C) phase boundary.
D) phase diagram.
E) transition state.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Iron crystallizes in the body-centered cubic lattice. What is the coordination number for Fe?
A) 4
B) 6
C) 8
D) 10
E) 12
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The strongest intermolecular interactions between hydrogen fluoride (HF) molecules arise from
A) dipole-dipole forces.
B) London dispersion forces.
C) hydrogen bonding.
D) ion-dipole interactions.
E) ionic bonds.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which arrow represents the process of vaporization?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The atomic planes in a graphite crystal are separated by 335 pm. At what angle would you find the first-order (n = 1) diffraction of 0.154-nm X rays from a graphite crystal?
A) 27.4°
B) 66.8°
C) 0.232°
D) 13.3°
E) 38.0°
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following substances crystallizes as a molecular solid?
A) KI
B) SiO2
C) Sn
D) CH3OH
E) Al2(SO4) 3
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
Variable melting point, variable hardness, and good conductor of heat and electricity are properties which describe a(n) ________ crystal.
Correct Answer

verified
Correct Answer
verified
True/False
In the packing of identical atoms with cubic unit cells, the packing efficiency increases as the coordination number increases.
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
________ ________ is a special type of dipole-dipole interaction.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the graph of vapor pressure to determine the normal boiling point of O2. 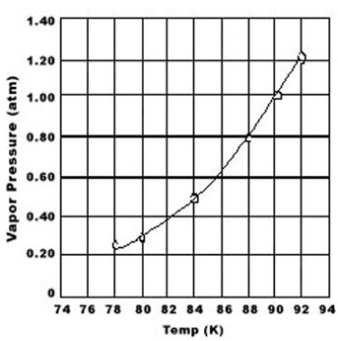
A) 92 K
B) 90. K
C) 88 K
D) 84 K
E) 78 K
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which substance has the highest vapor pressure at room temperature?
A) HF
B) HCl
C) HBr
D) HI
E) All of these substances have the same vapor pressure at room temperature.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 137
Related Exams