A) 205.1 nm
B) 384.6 nm
C) 683.8 nm
D) 1282 nm
E) > 1500 nm
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
According to ________, no two electrons in an atom can have the same four quantum numbers.
Correct Answer

verified
the Pauli ...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
The maximum number of electrons that can occupy an energy level described by the principal quantum number, n, is
A) n.
B) n + 1.
C) 2n.
D) 2n2.
E) n2.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Essay
In not more than three lines for each answer, briefly outline one important scientific contribution of each of the following. a) Planck b) De Broglie c) Heisenberg
Correct Answer

verified
a. Planck is responsible for first propo...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
If one s orbital were combined with one p orbital (dumbbell shaped) , which would best describe the resulting shape?
A) spherical
B) mostly spherical, slightly dumbbell
C) an equal ratio of spherical and dumbbell
D) slightly spherical, mostly dumbbell
E) mostly dumbbell
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of orbitals in a d subshell is
A) 1.
B) 2.
C) 3.
D) 5.
E) 7.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
________ is directly proportional to the frequency of a wave.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
________ is the wavelength associated with a moving particle.
A) The de Broglie wavelength
B) The Heisenburg wavelength
C) The Hund wavelength
D) The Aufbau wavelength
E) None of the answers is correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When a solid is heated, it emits electromagnetic radiation known as ________. An example of such radiation is the element of a stove stop burning bright red.
A) isoelectric radiation
B) visible and ultraviolet radiation
C) blackbody radiation
D) paramagnetic radiation
E) whitebody radiation
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the electron configuration for bromine?
A) 1s22s22p63s23p64s14d104p6
B) 1s22s22p63s23p64s24d104p5
C) 1s22s22p63s23p64s13d104p6
D) 1s22s22p63s23p64s23d104p4
E) 1s22s22p63s23p64s23d104p5
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to scientist James Clerk Maxwell in the year 1873, a(n) ________ consists of an electric field component and a magnetic field component.
A) electrostatic wave
B) isoelectric wave
C) atom
D) paramagnetic wave
E) electromagnetic wave
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which element has the following ground-state electron configuration? 1s2 2s2 2p6 3s2
A) Na
B) Mg
C) Al
D) Si
E) Ne
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A possible set of quantum numbers for the last electron added to complete an atom of gallium (Ga) in its ground state is 
A) A
B) B
C) C
D) D
E) E
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the quantum mechanical treatment of the hydrogen atom, which one of the following combinations of quantum numbers is not allowed? 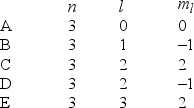
A) A
B) B
C) C
D) D
E) E
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the energy in joules of a mole of photons associated with red light of wavelength 7.00 × 102 nm? (c = 3.00 × 108 m/s; h = 6.63 × 10-34 J • s; NA = 6.022 × 1023 /mole)
A) 256 kJ
B) 1.71 × 105 J
C) 4.72 × 10-43 J
D) 12.4 kJ
E) 2.12 × 1042 J
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The ground-state electron configuration for an atom of indium is
A) [Kr]5s24p64d5.
B) [Ar]4s23d104p1.
C) [Ar]4s24p63d5.
D) [Kr]5s25p64d5.
E) [Kr]5s24d105p1.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
________ is the number of subshells in the shell designated as n = 2.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the wavelength of light having a frequency of 4.8 × 1014 s-1? (c = 3.00 x 108 m/s)
A) 0.0016 nm
B) 1600 m
C) 630 nm
D) 1600 nm
E) 6.3 × 10-7 nm
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
An electron in a 3p orbital could have a value of 2 for its angular momentum quantum number (l).
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Each shell (principal energy level) of quantum number n contains n subshells.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 135
Related Exams