A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following two representations of chemical reactions. Each sphere represents 1 mol of atoms. 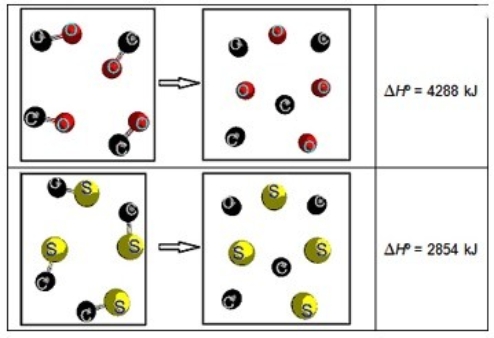 What is ΔHºrxn for the following reaction? CS(g) + O(g) → CO(g) + S(g)
What is ΔHºrxn for the following reaction? CS(g) + O(g) → CO(g) + S(g)
A) -359 kJ/mol
B) 1434 kJ/mol
C) -7142 kJ/mol
D) -1434 kJ/mol
E) 7142 kJ/mol
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ethanol, C2H5OH, is promoted as a clean fuel and is used as an additive in many gasoline mixtures. Calculate the ΔH°rxn for the combustion of ethanol. 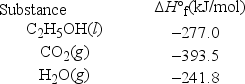
A) -1235.4 kJ
B) -751.8 kJ
C) -358.3 kJ
D) 358.3 kJ
E) 1235.4 kJ
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sand is converted to pure silicon in a three-step process. The final step in this process is as follows. SiCl4(g) + 2Mg(s) → 2MgCl(s) + Si(s) ΔH°rxn = -625.6 kJ/mol What is the enthalpy change if 25.0 mol of silicon tetrachloride is converted to elemental silicon?
A) -25.0 kJ
B) -626 kJ
C) -1.56 × l04 kJ
D) -3.13 × 104 kJ
E) -3.13 × 103 kJ
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
To which one of the following reactions, occurring at 25oC, does the symbol ΔHof [H2SO4(l) ] refer?
A) H2(g) + S(s) + 2 O2(g) <-----> H2SO4(l)
B) H2SO4(l) <-----> H2(g) + S(s) + 2 O2(g)
C) H2(g) + S(g) + 2 O2(g) <-----> H2SO4(l)
D) H2SO4(l) <-----> 2 H(g) + S(s) + 4 O(g)
E) 2 H(g) + S(g) + 4 O(g) <-----> H2SO4(l)
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the standard enthalpy change for the reaction 2C8H18(l) + 21O2(g) → 8CO(g) + 8CO2(g) + 18H2O(l) . Given: 2C8H18(l) + 25O2(g) → 16CO2(g) + 18H2O(l) ΔH° = -11,020. kJ/mol 2CO(g) + O2(g) → 2CO2(g) ΔH° = -566.0 kJ/mol
A) 1.0454 × 104 kJ/mol
B) -8756 kJ/mol
C) 1.1586 × 104 kJ/mol
D) -6492 kJ/mol
E) -1.0454 × 104 kJ/mol
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How much heat is released if 35.0 g of ethanol (C2H5OH) burns in excess oxygen? C2H5OH(l) + 3O2(g) → 2CO2(g) + 3H2O(l) ΔH°rxn = -1367 kJ/mol
A) 1797 kJ
B) 1367 kJ
C) 9.61 × 10-4 kJ
D) 4.78 × 104 kJ
E) 1040 kJ
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Chemical reactions in a bomb calorimeter occur at constant pressure.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which relationship is always correct?
A) potential energy + kinetic energy = constant
B) Δ = q + w
C) ΔU = ΔH - PΔV
D) H = U + PV
E) ΔH = qV
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following processes always results in an increase in the energy of a system?
A) The system loses heat and does work on the surroundings.
B) The system gains heat and does work on the surroundings.
C) The system loses heat and has work done on it by the surroundings.
D) The system gains heat and has work done on it by the surroundings.
E) None of these is always true.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is ΔH°rxn for the following reaction? 2H2O2(l) → 2H2O(l) + O2(g) ΔH°f(H2O(l) ) = -285.8 kJ/mol, ΔH°f(H2O2(l) ) = -187.6 kJ/mol
A) -196.4 kJ/mol
B) 98.2 kJ/mol
C) -98.2 kJ/mol
D) -473.4 kJ/mol
E) -946.8 kJ/mol
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
________ is the amount of heat required to raise the temperature of one gram of a substance by one degree Celsius.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which process is ΔH = ΔU?
A) Two moles of ammonia gas are cooled from 325°C to 300°C at a constant pressure of 1.2 atm.
B) One gram of water is vaporized at 100°C and 1 atm.
C) Two moles of hydrogen iodide gas react to form hydrogen gas and iodine gas in a 40-L container.
D) Solid calcium carbonate is heated to form solid calcium oxide and carbon dioxide gas in a container with variable volume.
E) One mole of solid carbon dioxide sublimes to the gas phase.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is ΔH°rxn for the following reaction? 2C8H18(l) + 17O2(g) → 16CO(g) + 18H2O(l) 2C8H18(l) + 25O2(g) → 16CO2(g) + 18H2O(l) ΔH°rxn = -11020. kJ/mol 2CO(g) + O2(g) → 2CO2(g) ΔH°rxn = -566.0 kJ/mol
A) -11020. kJ/mol
B) +6492 kJ/mol
C) -1964 kJ/mol
D) -6492 kJ/mol
E) -11586 kJ/mol
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is ΔH°rxn for the decomposition of calcium carbonate to calcium oxide and carbon dioxide? CaCO3(s) → CaO(s) + CO2(g) 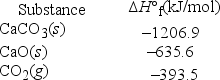
A) -2236.0 kJ/mol
B) -1449.0 kJ/mol
C) -177.8 kJ/mol
D) 177.8 kJ/mol
E) 2236.0 kJ/mol
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Octane (C8H18) undergoes combustion according to the following thermochemical equation. 2C8H18(l) + 25O2(g) → 16CO2(g) + 18H2O(l) ΔH°rxn = -1.0940 × 104 kJ/mol What is the standard enthalpy of formation of liquid octane? ΔH°f(CO2(g) ) = -393.5 kJ/mol and ΔH°f(H2O(l) ) = -285.8 kJ/mol
A) -250 kJ/mol
B) -10,940. kJ/mol
C) -2188 kJ/mol
D) -495 kJ/mol
E) 495 kJ/mol
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a particular process, 28 kJ of heat is absorbed and 15 kJ of work is done on the surroundings. What is ΔU?
A) ΔU = +43 kJ
B) ΔU = -43 kJ
C) ΔU = -13 kJ
D) ΔU = +13 kJ
E) ΔU = +28 kJ
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For which reaction is ΔH approximately (or exactly) equal to ΔU?
A) H2(g) + Br2(g) → 2HBr(g)
B) H2O(l) → H2O(g)
C) CaCO3(s) → CaO(s) + CO2(g)
D) 2H(g) + O(g) → H2O(l)
E) CH4(g) + 2O2(g) → CO2(g) + 2H2O(l)
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If 325 g of water at 4.2°C absorbs 12.28 kJ of heat, what is the final temperature of the water? The specific heat of water is 4.184 J/g·°C.
A) 4.21°C
B) 4.8°C
C) 9.0°C
D) 13.2°C
E) 2938°C
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When 0.560 g of Na(s) reacts with excess F2(g) to form NaF(s) , 13.8 kJ of heat is evolved at standard-state conditions. What is the standard enthalpy of formation (ΔH°f) of NaF(s) ?
A) 567 kJ/mol
B) -24.8 kJ/mol
C) -7.8 kJ/mol
D) 24.8 kJ/mol
E) -567 kJ/mol
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 141
Related Exams