A) [3,3]
B) [1,3]
C) [3,1]
D) [1,5]
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following polymers can be made at least in part from glucose derived from a renewable plant source?
A) Sorona
B) Polypropylene
C) Polyethylene
D) Teflon
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What type of sigmatropic rearrangement is illustrated below? ![What type of sigmatropic rearrangement is illustrated below? A) [1,5] B) [1,3] C) [1,4] D) [3,3]](https://d2lvgg3v3hfg70.cloudfront.net/TBMG1035/11ee4665_5cf7_5bfb_862d_dde9c93157c1_TBMG1035_00.jpg)
A) [1,5]
B) [1,3]
C) [1,4]
D) [3,3]
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is the Diels-Alder reaction called a thermal [4+2] cycloaddition?
A) Because the reaction is initiated by light; the dienophile has fourp electrons and the diene has twop electrons.
B) Because the reaction is initiated by heat; the diene has fourp electrons and the dienophile has twop electrons.
C) Because the reaction is initiated by light; the diene has fourp electrons and the dienophile has twop electrons.
D) Because the reaction is initiated by heat; the dienophile has fourp electrons and the diene has twop electrons.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about ap-bonding molecular orbital is true?
A) Ap-bonding molecular orbital is formed when twop orbitals of opposite phase overlap.
B) Ap-bonding molecular orbital is lower in energy than as-bonding molecular orbital.
C) Both the statements ap-bonding molecular orbital is formed when twop orbitals of similar phase overlap and ap-bonding molecular orbital is lower in energy than as-bonding molecular orbital are true.
D) Ap-bonding molecular orbital is formed when twop orbitals of similar phase overlap.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following reaction sequence. What is the correct classification of the first reaction in the sequence? ![Consider the following reaction sequence. What is the correct classification of the first reaction in the sequence? A) [3,3] Sigmatropic rearrangement B) [1,3] Sigmatropic rearrangement C) [1,5] Sigmatropic rearrangement D) [5,5] Sigmatropic rearrangement](https://d2lvgg3v3hfg70.cloudfront.net/TBMG1035/11ee4665_5cf7_830c_862d_85158e53b564_TBMG1035_00.jpg)
A) [3,3] Sigmatropic rearrangement
B) [1,3] Sigmatropic rearrangement
C) [1,5] Sigmatropic rearrangement
D) [5,5] Sigmatropic rearrangement
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about the Cope rearrangement isnot true?
A) The Cope rearrangement involves the rearrangement of a 1,5-diene to an isomeric 1,5-diene.
B) The Cope rearrangement takes place readily in a suprafacial pathway under photochemical conditions.
C) The Cope rearrangement involves three electron pairs; two inp bonds and one in as bond.
D) The Cope rearrangement takes place readily in a suprafacial pathway, when heated.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about ap* antibonding molecular orbital is true?
A) Ap* antibonding molecular orbital is formed when twop orbitals of similar phase overlap.
B) Both the statements ap* antibonding molecular orbital is formed when twop orbitals of opposite phase overlap and ap* antibonding molecular orbital is a higher-energy molecular orbital than ap bonding molecular orbital are true.
C) Ap* antibonding molecular orbital is formed when twop orbitals of opposite phase overlap.
D) Ap* antibonding molecular orbital is a higher-energy molecular orbital than ap bonding molecular orbital.
F) B) and C)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Which of the following statements about sigmatropic reactions isnot true?
A) Thep bonds rearrange in a sigmatropic reaction.
B) A sigmatropic reaction is an intramolecular pericyclic reaction.
C) In a sigmatropic reaction,s bond is broken in one of the reactants.
D) The number ofp bonds in the reactants and product differs in a sigmatropic reaction.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about an electrocyclic ring-opening reaction isnot true?
A) The product of an electrocyclic ring-opening reaction contains one fewerp bond than the reactant.
B) The product of an electrocyclic ring-opening reaction contains one morep bond than the reactant.
C) An electrocyclic ring-opening reaction requires a source of energy (heat or light) .
D) An electrocyclic ring-opening reaction is an intramolecular reaction.
F) None of the above
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Which of the following statements about the Claisen rearrangement is true?
A) The Claisen rearrangement occurs readily in a suprafacial pathway under photochemical conditions.
B) The Claisen rearrangement involves three electron pairs; two inp bonds and one in as bond.
C) The Claisen rearrangement occurs readily in an antarafacial pathway under thermal conditions.
D) The Claisen rearrangement involves the rearrangement of an unsaturated ether to a b,g-unsaturated carbonyl compound.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about thermal electrocyclic reactions isnot true?
A) The number ofs bonds in the conjugated polyene determines whether rotation is conrotatory or disrotatory.
B) In thermal reactions, we consider the orbitals of the HOMO of the ground state electronic configuration to determine the course of the reaction.
C) Thermal electrocyclic reactions occur in a conrotatory fashion for a conjugated polyene with an even number ofp bonds.
D) Thermal electrocyclic reactions occur in a disrotatory fashion for a conjugated polyene with an odd number ofp bonds.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following polymers can be recycled into floor mats?
A) LDPE
B) PVC
C) HDPE
D) PET
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What type of sigmatropic rearrangement is illustrated below? ![What type of sigmatropic rearrangement is illustrated below? A) [3,3] B) [1,4] C) [1,5] D) [1,3]](https://d2lvgg3v3hfg70.cloudfront.net/TBMG1035/11ee4665_5cf7_5bfa_862d_db5d716529dd_TBMG1035_00.jpg)
A) [3,3]
B) [1,4]
C) [1,5]
D) [1,3]
F) B) and C)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
What type of sigmatropic rearrangement is illustrated below? ![What type of sigmatropic rearrangement is illustrated below? A) [1,3] B) [1,4] C) [1,5] D) [3,3]](https://d2lvgg3v3hfg70.cloudfront.net/TBMG1035/11ee4665_5cf6_e6c9_862d_bb87ea604df2_TBMG1035_00.jpg)
A) [1,3]
B) [1,4]
C) [1,5]
D) [3,3]
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Bakelite is formed by the acid-catalyzed polymerization of phenol with formaldehyde. What is (are) the product(s) of the first step in this polymerization, shown below? (Note: in the answers below the hydroxymethyl group is -CH2OH.) 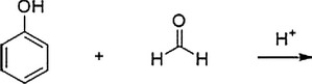
A) Ortho andpara-hydroxybenzaldehyde
B) Meta-(hydroxymethyl) phenol
C) Ortho andpara-(hydroxymethyl) phenol
D) Meta-hydroxybenzaldehyde
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about cycloaddition reactions isnot true?
A) Cycloaddition reactions are stereospecific.
B) Cycloaddition reactions are concerted.
C) The course of the reaction is determined by the symmetry of the molecular orbitals of the products.
D) Cycloaddition reactions form a cyclic product with two news bonds.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about orbital symmetry and cycloaddition reactions is true?
A) Thermal cycloadditions involving an odd number ofp bonds proceed by an antarafacial pathway.
B) Thermal cycloadditions involving an even number ofp bonds proceed by a suprafacial pathway.
C) Photochemical cycloadditions involving an even number ofp bonds proceed by an antarafacial pathway.
D) Photochemical cycloadditions involving an even number ofp bonds proceed by a suprafacial pathway.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about photochemical electrocyclic reactions isnot true?
A) Photochemical electrocyclic reactions occur in a disrotatory fashion for a conjugated polyene with an even number ofp bonds.
B) Photochemical electrocyclic reactions occur in a conrotatory fashion for a conjugated polyene with an odd number ofp bonds.
C) In photochemical reactions, we consider the orbitals of the HOMO of the excited state to determine the course of the reaction.
D) In photochemical reactions, we consider the orbitals of the LUMO of the excited state to determine the course of the reaction.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about orbital symmetry and cycloaddition reactions is true?
A) Photochemical sycloadditions involving an even number ofp bonds proceed by an antarafacial pathway.
B) Photochemical cycloadditions involving an odd number ofp bonds proceed by a suprafacial pathway.
C) Thermal cycloadditions involving an even number ofp bonds proceed by a suprafacial pathway.
D) Thermal cycloadditions involving an odd number ofp bonds proceed by a suprafacial pathway.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 23
Related Exams