A) Two different aldehydes with a-hydrogens are able to form a single aldol condensation product.
B) Any pair of aldehyde or ketone reactants where one of the reactants has no a-hydrogens will lead to a single aldol product.
C) Two different ketones with a-hydrogens are able to form a single aldol condensation product.
D) Any aldehyde and ketone mixed together can react to form a single condensation product.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major product of the following reaction? 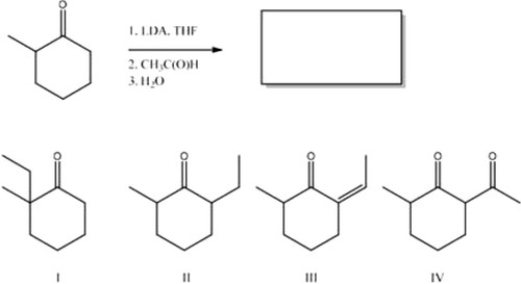
A) II
B) IV
C) III
D) I
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If a Claisen condensation reaction is run using methyl propanoate as the reactant, NaOCH3 is the ideal base. Why is it important to use NaOCH3 and not NaOCH2CH3?
A) NaOCH3 is a weaker base than NaOCH2CH3, and this reaction requires a weaker base.
B) Transesterfication can occur when esters react, and this transesterfication would result in a mixture of products.
C) NaOCH3 is more soluble than NaOCH2CH3 in organic solvents, and this reaction requires a full equivalent of base to proceed. A full equivalent of NaOCH2CH3 would not dissolve, so the reaction would not proceed.
D) NaOCH3 is a stronger base than NaOCH2CH3, and this reaction requires a stronger base.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major product of the following reaction? 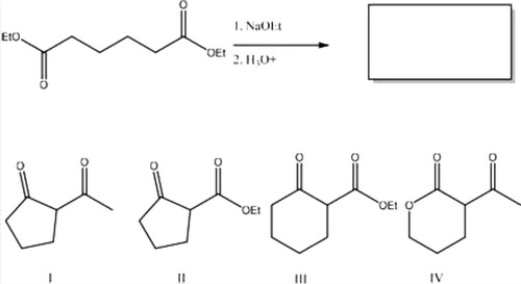
A) II
B) IV
C) I
D) III
F) B) and D)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
In what situation can the yield of a single crossed Aldol product be increased?
A) The electrophilic carbonyl component is relatively unhindered and is used in excess.
B) The nucleophilic carbonyl component is relatively unhindered and is used in excess.
C) The electrophilic carbonyl carbon component is relatively hindered and is used in limited amount.
D) The nucleophilic carbonyl component is relatively hindered and is used in limited amount.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major product of the following reaction? 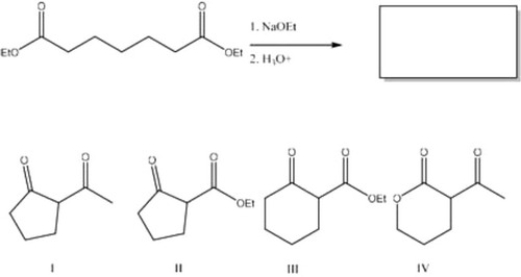
A) IV
B) II
C) III
D) I
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following reaction is an example of what type of reaction? 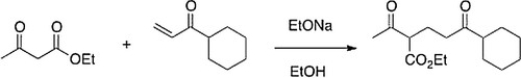
A) Dieckmann condensation
B) Robinson annulation
C) Aldol self-condensation
D) Michael reaction
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Complete this statement: A major difference between the Aldol condensation and the Claisen condensation reactions is that
A) the Aldol reaction is base catalyzed while the Claisen reaction is acid-catalyzed.
B) the Aldol reaction is base catalyzed while the Claisen reaction requires a full equivalent of base.
C) the Aldol reaction involves substitution while the Claisen reaction involves addition.
D) the Aldol reaction is acid catalyzed while the Claisen reaction is base-catalyzed.
F) A) and B)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Would this crossed Aldol reaction work well? Why or why not? 
A) No, the aldehyde is significantly more acidic, so this enolate cannot be formed selectively.
B) No, the diketone is significantly more acidic, so this enolate cannot be formed selectively.
C) Yes, the diketone is significantly more acidic, so this enolate can be formed selectively.
D) Yes, the aldehyde is significantly more acidic, so this enolate can be formed selectively.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Under basic conditions, the Aldol reaction is reversible, but dehydration is not. What is the reason for this difference in reactivity?
A) The initial Aldol product is an alkoxide, so the reaction is energetically downhill going toward the product.
B) The initial Aldol product is an alkoxide, so the reaction is energetically downhill going toward the starting materials.
C) Water is a stable molecule.
D) The initial Aldol product is an alkoxide, so the reaction is not energetically downhill in either direction.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The b-hydroxy carbonyl product of an Aldol reaction is oftentimes not the final isolated product; what is the explanation for this result?
A) The hydroxy group reacts with the carbonyl to form a ketal.
B) The hydroxy group is oxidized to a carbonyl.
C) Hydroxide is eliminated via an enolate intermediate.
D) It undergoes elimination, since water is a good leaving group.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about Aldol reactions with either aldehydes or ketones is true?
A) Equilibrium favors the products with aldehydes; equilibrium favors the starting materials with ketones.
B) Equilibrium favors the starting materials with aldehydes; equilibrium favors the products with ketones.
C) Equilibrium favors the starting materials with both aldehydes and ketones.
D) Equilibrium favors the products with both aldehydes and ketones.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A key step in the biosynthesis of glucose involves the reaction of glyceraldehyde 3-phosphate with dihydroxyacetone phosphate to form fructose 1,6-bisphosphate in the presence of an aldolase enzyme. The end product formed from this reaction is
A) a single form of fructose in the cyclic hemiacetal form.
B) a form of critic acid.
C) a single form of fructose in the acyclic form.
D) an equilibrium between an acyclic form of fructose and a cyclic hemiacetal.
F) A) and C)
Correct Answer

verified
D
Correct Answer
verified
Showing 1 - 13 of 13
Related Exams