A) Histidine
B) Isoamyl acetate
C) Ethyl acetoacetate
D) Pyridine
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following molecule is called 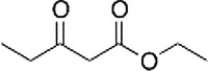
A) an ethyl acetoacetate.
B) a 1,3-diketopentanoate.
C) a b-keto ester.
D) a diethyl malonate.
F) None of the above
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 
A) II
B) I
C) IV
D) III
F) None of the above
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Starting with cyclohexanone, how could you prepare the diketone below? 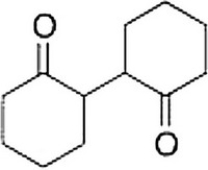
A) Convert cyclohexanone into the a-bromoketone and then react this with the enolate of cyclohexanone.
B) Convert cyclohexanone into an enamine with diethylamine and then react this with more cyclohexanone.
C) Treat cyclohexanone with a base under thermodynamic conditions.
D) Hydrogenate cyclohexanone with Raney nickel.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major product of the following reaction? 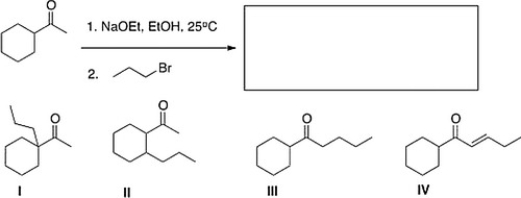
A) IV
B) I
C) III
D) II
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compound would most likely undergo racemization under basic conditions?
A) (S) -4-Iodo-2-pentanone
B) (R) -5-Iodo-5-bromo-2-pentanone
C) (R) -3-Iodo-2-pentanone
D) (R) -4-Iodo-2-pentanone
F) B) and C)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Which of the following is the least acidic compound? 
A) II
B) I
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the appropriate sequence of reactions to accomplish the following synthesis. ![Select the appropriate sequence of reactions to accomplish the following synthesis. A) [1] LDA; [2] BrCH<sub>2</sub>CH<sub>2</sub>CH<sub>2</sub>CH<sub>2</sub>Br; [3] NaOEt B) [1] Br<sub>2</sub>, CH<sub>3</sub>CO<sub>2</sub>H; [2] Li<sub>2</sub>CO<sub>3</sub>, LiBr, DMF; [3] CH<sub>3</sub>CH<sub>2</sub>CH<sub>2</sub>CH<sub>2</sub>Br C) [1] Br<sub>2</sub>, CH<sub>3</sub>CO<sub>2</sub>H; [2] Mg, Et<sub>2</sub>O; [3] CH<sub>3</sub>CH<sub>2</sub>CH<sub>2</sub>CH<sub>2</sub>Br D) [1] NaOEt; [2] BrCH<sub>2</sub>CH<sub>2</sub>CH<sub>2</sub>CH<sub>2</sub>Br; [3] LDA](https://d2lvgg3v3hfg70.cloudfront.net/TBMG1035/11ee4665_5cff_24aa_862d_cbbaf637032e_TBMG1035_00.jpg)
A) [1] LDA; [2] BrCH2CH2CH2CH2Br; [3] NaOEt
B) [1] Br2, CH3CO2H; [2] Li2CO3, LiBr, DMF; [3] CH3CH2CH2CH2Br
C) [1] Br2, CH3CO2H; [2] Mg, Et2O; [3] CH3CH2CH2CH2Br
D) [1] NaOEt; [2] BrCH2CH2CH2CH2Br; [3] LDA
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the most acidic compound? 
A) I
B) III
C) IV
D) II
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 
A) IV
B) I
C) III
D) II
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction below is a direct enolate alkylation. It has been found that this reaction only works well with unhindered methyl and 1° alkyl halides. Pick the statement that best explains this observation. 
A) Hindered alkyl halides do not undergo SN1 reactions.
B) The nucleophilic enolate requires a reaction center that has a positive charge.
C) Hindered alkyl halides do not undergo SN2 reactions.
D) Methyl and 1° alkyl halides can form carbocations that can readily react with the nucleophilic enolate.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Treatment of 2-hexanone with NaOCH2CH3 followed by CH3Br affords compound X (C7H14O) as the major product. X shows a strong absorption in the IR spectrum at 1713 cm?1, and its 1H NMR data is given below. What is the structure of X?
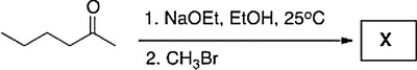
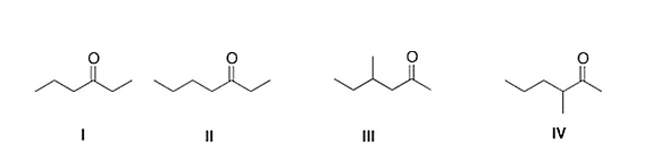
A) IV
B) III
C) II
D) I
F) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 1 - 12 of 12
Related Exams