A) 1-chloro-1-butene
B) 1-chloro-2-butene
C) 2-chloro-1-butene
D) ethyl chloride + acetylene
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The formula CH3C CCH3 represents
A) an alkane.
B) a cycloalkane.
C) an alkene.
D) an alkyne.
E) an aromatic compound.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Water and ethylene are combined at high temperature and pressures in the presence of sulfuric acid to produce ethanol according to the following reaction: CH2=CH2(g) + H2O(g) CH3CH2OH(g) What is the hybridization of the bolded carbon atoms in the reactants and products, respectively
A) sp3, sp2
B) sp2, sp3
C) sp, sp2
D) sp3, sp
E) sp2, sp2
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following functional groups is found in carboxylic acids 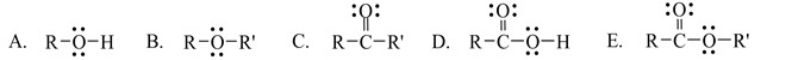
A) A
B) B
C) C
D) D
E) E
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which option represents dimethyl ether
A) CH3CH2OH
B) CH3OH
C) CH3COCH3
D) CH3OCH3
E) CH3CH2OCH3
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The expected product from the addition of HCl to CH3-CH2-CH=CH2 is
A) CH3-CH2-CH=CHCl
B) CH3-CH2-CCl=CH2
C) CH3-CHCl-CH=CH2
D) CH3-CH2-CH2-CH2Cl
E) CH3-CH2-CHCl-CH3
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these reactions leads to a change in the hybridization of one or more carbon atoms
A) free radical halogenation of an alkane
B) hydrolysis of an ester to yield an acid and an alcohol
C) substitution of an aromatic ring using a halogen
D) oxidation of an alcohol to yield a carboxylic acid
E) neutralization of an amine using a strong mineral acid
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The systematic name for the hydrocarbon with the following structural formula is 1-ethyl-2-methylbutane. 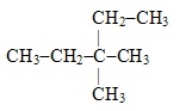
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The formulas for the alcohol and the carboxylic acid (respectively) from which the following ester may be synthesized are
CH3-(CH2)14-COOH and CH3-(CH2)9-OH. 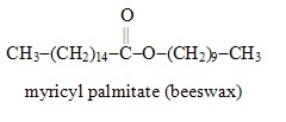
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The systematic name for the compound with the following structural formula is 4,5-dimethyl-2-hexene. 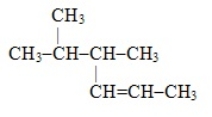
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following functional groups is found in alcohols. 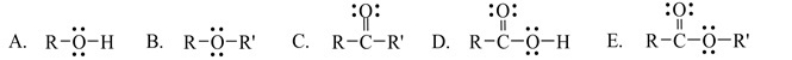
A) A
B) B
C) C
D) D
E) E
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The correct structure for 2,3,3-trimethylpentane is
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction of ethylene and water yields
A) an aldehyde.
B) an ester.
C) an alcohol.
D) an ether.
E) an organic acid.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these pairs are geometric isomers
A) CH3CH2-O-CH2CH3 and CH3CH2CH2CH2OH
B) ![]()
C) ![]()
D) ![]()
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The alkane with six carbon atoms is called
A) butane.
B) hexane.
C) heptane.
D) butene.
E) none of these.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Amines are
A) organic bases that react with water to produce ammonia.
B) organic acids that react with water to produce ammonia.
C) organic bases that react with acids to form ammonium salts.
D) organic acids that react with bases to form ammonium salts.
E) None of the above.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these is the systematic name for the compound represented below 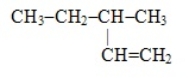
A) 2-ethylbutane
B) 3-methylpentene
C) 3-methyl-1-pentene
D) 3-methyl-1-hexene
E) 2-methylhexane
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Bromination of benzene (C6H6) , an aromatic compound,
A) occurs by substitution rather than addition.
B) occurs by addition rather than substitution.
C) occurs more rapidly than bromination of a nonaromatic compound.
D) results in formation of 1,2,3,4,5,6-hexabromocyclohexane.
E) occurs in the absence of a catalyst.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name of the molecule CH3CH2CH2CH2CH2CH2CH2CH3
A) butane
B) hexane
C) heptane
D) octane
E) nonane
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Name the following compound: 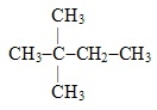
A) 2,2-dimethylbutane
B) 3,3-dimethylbutane
C) 2,2-dimethylpropane
D) 2-methyl,2-ethyllpropane
E) None of the above
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 66
Related Exams