A) Red
B) Yellow
C) Green
D) Indigo
E) Violet
G) None of the above
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Which element has the following ground-state electron configuration [Kr]5s24d105p3
A) Sn
B) Sb
C) Pb
D) Bi
E) Te
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The electron configuration of a ground-state copper atom is
A) [Ar]4s24d4.
B) [Ar]4s24p63d3.
C) [Ar]4s23d9.
D) [Ar]3d9.
E) [Ar]4s13d10.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the following diagram of a wave 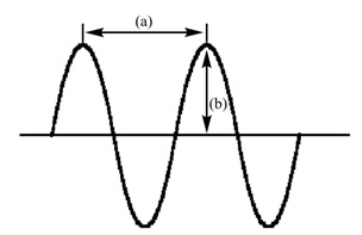
A) (a) is amplitude and (b) is wavelength
B) (a) is frequency and (b) is amplitude
C) (a) is wavelength and (b) is frequency
D) (a) is amplitude and (b) is frequency
E) (a) is wavelength and (b) is amplitude
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The colors of the visible spectrum are blue, green, orange, red, violet, and yellow. Of these colors, _______ has the most energy.
A) Red
B) Yellow
C) Green
D) Indigo
E) Violet
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following, if any, is the correct ground state electron configuration for an iodine atom
A) [Kr] 5s24d105p5 or 1s22s22p63s23p64s23d104p65s24d105p5
B) [Kr] 5s24d105p4 or 1s22s22p63s23p64s23d104p65s24d105p4
C) [Kr] 5s24d105p3 or 1s22s22p63s23p64s23d104p65s24d105p3
D) [Kr] 5s24d105p2 or 1s22s22p63s23p64s23d104p65s24d105p2
E) None of the above
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
List the following sets of quantum numbers in order of increasing energy: I. n = 4, l = 1, ml = 1, ms = +1/2 II. n = 3, l = 2, ml = -1, ms = +1/2 III. n = 4, l = 0, ml = 0, ms = +1/2
A) I < II < III
B) II < III < I
C) III < II < I
D) I < III < II
E) III < I < II
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A single pulse of a laser yields an average of 5.00 * 1018 photons with l= 633 nm. If melting ice to water at 0 C requires 6.01 kJ/mol, what is the fewest number of laser pulses needed to melt 10.0 g of ice
A) 38300
B) 3830
C) 3340
D) 2120
E) 212
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which ground-state atom has an electron configuration described by the following orbital diagram 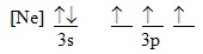
A) phosphorus
B) nitrogen
C) arsenic
D) vanadium
E) none of these
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The ground-state electron configuration for an atom of indium is
A) [Kr]5s24p64d5.
B) [Ar]4s23d104p1.
C) [Ar]4s24p63d5.
D) [Kr]5s25p64d5.
E) [Kr]5s24d105p1.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
For all atoms of the same element, the 2s orbital is larger than the 1s orbital.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following, if any, is the correct ground state electron configuration for a lead atom
A) [Xe] 6s24f145d106p1 or 1s22s22p63s23p64s23d104p65s24d105p66s24f145d106p1
B) [Xe] 6s24f145d86p2 or 1s22s22p63s23p64s23d104p65s24d105p66s24f145d86p2
C) [Xe] 6s24f145d106p2 or 1s22s22p63s23p64s23d104p65s24d105p66s24f145d106p2
D) [Kr] 6s24f145d106p2 or 1s22s22p63s23p64s23d104p65s24d105p66s24f145d106p2
E) None of the above
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following scientists made this contribution to quantum: "It is impossible to know simultaneously both the momentum and the position of a particle with certainty."
A) Planck
B) Bohr
C) Heisenberg
D) Einstein
E) Pauli
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The quantum numbers, n = 3, l = 1, ml = 0, ms = +1/2, represent an electron in a ____ subshell.
A) 3p
B) 3s
C) 3d
D) 3f
E) None of the above
G) A) and D)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Using the figure below, categorize electromagnetic radiation with an energy of 6.6 x 10-16 J/photon. 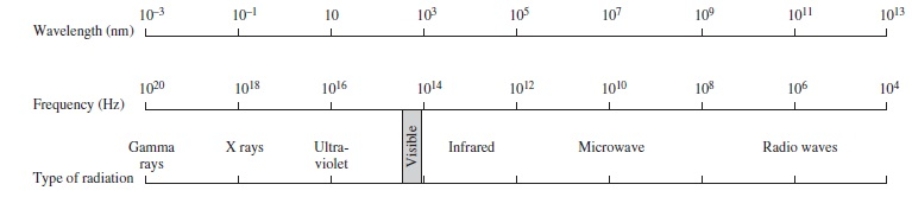
A) Gamma rays
B) X rays
C) Ultraviolet
D) Infrared
E) Microwave
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is diamagnetic both in its ground state and in all of its excited states
A) Mg
B) Ne
C) Cu
D) Zn
E) none of these
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the maximum number of electrons in an atom that can have the following quantum numbers N = 3 l = 1
A) 18
B) 6
C) 3
D) 2
E) 1
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the electron in a hydrogen atom falls from the n = 3 excited energy level to the ground state energy level, a photon with wavelength lis emitted. An electron having this same wavelength would have a velocity of
A) 7.10 * 103 m/s
B) 2.93 * 106 m/s
C) 2.93 * 103 m/s
D) 7.10 m/s
E) 3.00 * 108 m/s
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the frequency of visible light having a wavelength of 486 nm.
A) 2.06 * 1014 /s
B) 2.06 * 106 /s
C) 6.17 * 1014 /s
D) 1.20 * 10-15 /s
E) 4.86 * 10-7 /s
G) B) and E)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
How many unpaired electrons does an atom of sulfur have in its ground state
A) 0
B) 1
C) 2
D) 3
E) 4
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 135
Related Exams