A) 2
B) 7
C) 8
D) 16
E) None of these choices is correct.
G) C) and D)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Consider the following balanced redox reaction.3CuO(s) + 2NH3(aq) → N2(g) + 3H2O(l) + 3Cu(s) Which of the following statements is ?
A) CuO(s) is the oxidizing agent and copper is reduced.
B) CuO(s) is the oxidizing agent and copper is oxidized.
C) CuO(s) is the reducing agent and copper is oxidized.
D) CuO(s) is the reducing agent and copper is reduced.
E) CuO(s) is the oxidizing agent and N2(g) is the reducing agent.
G) C) and E)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Which electrochemical cell pictured below corresponds to the following cell diagram? Cr(s) | Cr3+(aq, 1.0 M) || Sn2+(aq, 1.0 M) | Sn(s)
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the following redox equation is balanced with the smallest whole number coefficients, what is the coefficient of Sn(OH) 3−? Bi(OH) 3(s) + Sn(OH) 3-(aq) → Sn(OH) 62−(aq) + Bi(s) (basic solution)
A) 1
B) 2
C) 3
D) 6
E) 12
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which element is associated with the term "galvanized"?
A) Ga
B) Zn
C) Cd
D) Hg
E) Pb
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Complete and balance the following redox equation.When properly balanced using the smallest whole-number coefficients, the coefficient of S is H2S + HNO3 → S + NO + H2O (acidic solution)
A) 1.
B) 2.
C) 3.
D) 5.
E) 6.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A voltaic cell is prepared using copper and silver.Its cell notation is shown below.Cu(s) | Cu2+(aq) || Ag+(aq) | Ag(s) Which reaction occurs at the cathode?
A) Cu(s) → Cu2+(aq) + 2e−
B) Cu2+(aq) + 2e−→ Cu(s)
C) Ag(s) → Ag+(aq) + e−
D) Ag+(aq) + e− → Ag(s)
E) Cu(s) + 2Ag+(aq) → Cu2+(aq) + 2Ag(s)
G) D) and E)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Consider the reaction CuO(s) + H2(g) → Cu(s) + H2O(l) In this reaction, which substances are the oxidizing agent and reducing agent, respectively?
A) CuO and H2
B) H2 and CuO
C) CuO and Cu
D) H2O and H2
E) Cu and H2O
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is not a redox reaction?
A) Al(OH) 4−(aq) + 4H+(aq) → Al3+(aq) + 4H2O(l)
B) C6H12O6(s) + 6O2(g) → 6CO2(g) + 6H2O(l)
C) Na6FeCl8(s) + 2Na(l) → 8NaCl(s) + Fe(s)
D) 2H2O2(aq) → 2H2O(l) + O2(g)
E) CO2(g) + H2(g) → CO(g) + H2O(g)
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Complete and balance the following redox equation using the set of smallest whole numbers coefficients.What is the sum of the coefficients? HI + HNO3 → I2 + NO (acidic solution)
A) 5
B) 7
C) 14
D) 17
E) None of these choices is correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the following redox equation is balanced with the smallest whole number coefficients, the coefficient for the hydrogen sulfate ion will be ______. Al(s) + HSO4−(aq) + OH−(aq) → Al2O3(s) + S2−(aq) + H2O(l)
A) 1
B) 3
C) 4
D) 6
E) 8
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A voltaic cell prepared using aluminum and nickel has the following cell notation.Al(s) | Al3+(aq) || Ni2+(aq) | Ni(s) Which reaction occurs at the anode?
A) Al(s) → Al3+(aq) + 3e−
B) Al3+(aq) + 3e− → Al(s)
C) Ni(s) → Ni2+(aq) + 2e−
D) Ni2+(aq) + 2e− → Ni(s)
E) Ni(s) + Ni2+(aq) → Al(s) + Al3+(aq)
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Aluminum does not corrode in the same manner as iron does, because
A) aluminum does not react with oxygen gas.
B) a protective layer of aluminum oxide forms on the metal surface.
C) aluminum is harder to oxidize than iron.
D) iron gives cathodic protection to aluminum.
E) the electrical circuit cannot be completed on an aluminum surface.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Complete and balance the following redox equation.Now sum the coefficients of all species in the balanced equation.(Remember to add the coefficients that are equal to one, and to add the coefficients of any species added to the equation.) The sum of the smallest whole number coefficients is Bi(OH) 3 + SnO22− → Bi + SnO32− (basic solution)
A) 32
B) 25
C) 16
D) 13
E) 4
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the following redox equation is balanced with the smallest whole number coefficients, what is the coefficient for nitrogen dioxide? I2(s) + HNO3(aq) → HIO3(aq) + NO2(g) + H2O(l)
A) 1
B) 2
C) 4
D) 5
E) 10
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the half-reaction at the anode in a lead storage battery?
A) Pb(s) + PbO2(s) + 4H+(aq) + 2SO42−(aq) → 2PbSO4(s) + 2H2O(l)
B) PbO2(s) + 4H+(aq) + 2SO42−(aq) + 2e− → PbSO4(s) + 2H2O(l)
C) Pb(s) + SO42-(aq) → PbSO4(s) + 2e−
D) Pb(s) → Pb(s) + 2e−
E) H2(g) → 2H+(aq) + 2e−
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the half-reaction at the cathode in a lead storage battery?
A) Pb(s) + PbO2(s) + 4H+(aq) + 2SO42-(aq) → 2PbSO4(s) + 2H2O(l)
B) PbO2(s) + 4H+(aq) + 2SO42−(aq) + 2e− → PbSO4(s) + 2H2O(l)
C) Pb(s) + SO42−(aq) → PbSO4(s) + 2e−
D) Pb(s) → Pb(s) + 2e−
E) H2(g) → 2H+(aq) + 2e−
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A certain electrochemical cell has for its cell reaction: Zn + HgO → ZnO + Hg Which is the half-reaction occurring at the anode?
A) HgO + 2e− → Hg + O2−
B) Zn2++ 2e− → Zn
C) Zn → Zn2+ + 2e−
D) ZnO + 2e− → Zn
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following redox equation. Mn(OH) 2(s) + MnO4−(aq) → MnO42−(aq) (basic solution) When the equation is balanced with the smallest whole number coefficients, what is the coefficient for OH−(aq) and on which side of the equation is OH−(aq) present?
A) 4, reactant side
B) 4, product side
C) 6, reactant side
D) 6, product side
E) 8, reactant side
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Based on the following electrochemical cell, which statement is ? 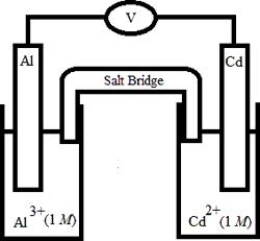 Half-Reaction E° (V) Al3+(aq) + 3e- →
−1) 66
Al(s)
Cd2+(aq) + 2e- →
−0) 40
Cd(s)
Half-Reaction E° (V) Al3+(aq) + 3e- →
−1) 66
Al(s)
Cd2+(aq) + 2e- →
−0) 40
Cd(s)
A) Al(s) is oxidized and is the anode.
B) Al(s) is oxidized and is the cathode.
C) Cd(s) is oxidized and is the anode.
D) Cd(s) is oxidized and is the cathode.
E) No reaction occurs because E°cell < 0.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 30
Related Exams