A) PbO
B) PbO2
C) Pb2O
D) PbO4
E) Pb2O3
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Essay
For each of the following names,write down the corresponding formula,including charge where appropriate (atomic numbers and mass numbers are not required): a.zinc ion b.nitrite ion c.carbonic acid d.cyanide ion
Correct Answer

verified
a.Zn2+
b.NO2-
...View Answer
Show Answer
Correct Answer
verified
b.NO2-
...
View Answer
Multiple Choice
Which one of the following statements about atoms and subatomic particles is correct?
A) Rutherford discovered the atomic nucleus by bombarding gold foil with electrons.
B) The proton and the neutron have identical masses.
C) The neutron's mass is equal to that of a proton plus an electron.
D) A neutral atom contains equal numbers of protons and electrons.
E) An atomic nucleus contains equal numbers of protons and neutrons.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Lead (Pb)is a main-group element.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Barium sulfate is used in manufacturing photographic paper.What is its formula?
A) BaSO4
B) Ba(SO4) 2
C) Ba2SO4
D) Ba2(SO4) 3
E) BaSO3
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements are the least reactive?
A) alkali metals
B) noble gases
C) halogens
D) alkaline earth metals
E) metalloids
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The substance,CaSe,is used in materials which are electron emitters.What is its name?
A) calcium monoselenide
B) calcium(II) selenide
C) calcium selenide
D) calcium(I) selenide
E) calcium(II) selenium
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Zinc acetate is used in preserving wood and in manufacturing glazes for porcelain.What is its formula?
A) ZnAc2
B) ZnCH3COO
C) Zn(CH3COO) 2
D) Zn2CH3COO
E) ZnCH3COCH3
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rutherford bombarded gold foil with alpha ( ) particles and found that a small percentage of the particles were deflected.Which of the following was not accounted for by the model he proposed for the structure of atoms?
A) the small size of the nucleus
B) the charge on the nucleus
C) the total mass of the atom
D) the existence of protons
E) the presence of electrons outside the nucleus
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Who is credited with first measuring the charge of the electron?
A) Dalton
B) Gay-Lussac
C) Thomson
D) Millikan
E) Rutherford
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ions occurs commonly?
A) N3+
B) S6+
C) O2-
D) Ca+
E) Cl+
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The compound, (NH4) 2S,can be used in analysis for trace amounts of metals present in a sample.What is its name?
A) ammonium sulfide
B) diammonium sulfide
C) ammonium sulfite
D) ammonia(I) sulfite
E) ammonium(I) sulfide
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Who is credited with discovering the atomic nucleus?
A) Dalton
B) Gay-Lussac
C) Thomson
D) Millikan
E) Rutherford
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a metalloid?
A) carbon,C,Z = 6
B) sulfur,S,Z = 16
C) germanium,Ge,Z = 32
D) iridium,Z = 77
E) bromine,Br,Z = 35
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ammonium sulfate, (NH4) 2SO4,is a fertilizer widely used as a source of nitrogen.Calculate its molecular mass.
A) 63.07 amu
B) 114.l0 amu
C) 118.13 amu
D) 128.11 amu
E) 132.13 amu
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Bromine is the only nonmetal that is a liquid at room temperature.Consider the isotope bromine-81, 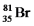 .Select the combination which lists the correct atomic number,neutron number,and mass number,respectively.
.Select the combination which lists the correct atomic number,neutron number,and mass number,respectively.
A) 35,46,81
B) 35,81,46
C) 81,46,35
D) 46,81,35
E) 35,81,116
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Who is credited with measuring the mass/charge ratio of the electron?
A) Dalton
B) Gay-Lussac
C) Thomson
D) Millikan
E) Rutherford
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following combinations of names and formulas of ions is incorrect?
A) O2- oxide
B) Al3+ aluminum
C) NO3- nitrate
D) PO43- phosphate
E) CrO42- chromate
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Iodine pentafluoride reacts slowly with glass and violently with water.Determine its molecular mass.
A) 653.52 amu
B) 259.89 amu
C) 221.90 amu
D) 202.90 amu
E) 145.90 amu
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Essay
a.Give the names of the following ions: (i)O22- (ii)SO42- b.Write down the formulas of the following ions: (i)ammonium (ii)nitrate
Correct Answer

verified
a.(i)perox...View Answer
Show Answer
Correct Answer
verified
View Answer
Showing 61 - 80 of 91
Related Exams