A) ( Hsoln > 0,entropy of system decreases)
B) ( Hsoln 0,entropy of system decreases)
C) ( Hsoln 0,entropy change of system 0)
D) ( Hsoln 0,entropy of system increases)
E) None of these choices is correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Colligative properties depend on
A) the chemical properties of the solute.
B) the chemical properties of the solvent.
C) the masses of the individual ions.
D) the molar mass of the solute.
E) the number of particles dissolved.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Heats of hydration may be either positive or negative.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Dimethylglyoxime,DMG,is an organic compound used to test for aqueous nickel(II) ions.A solution prepared by dissolving 65.0 g of DMG in 375 g of ethanol boils at 80.3°C.What is the molar mass of DMG? Kb = 1.22°C/m,boiling point of pure ethanol = 78.5°C
A) 44.1 g/mol
B) 65.8 g/mol
C) 117 g/mol
D) 131.6 g/mol
E) 553 g/mol
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the molarity of a solution prepared by diluting 1.85 L of 6.5 M KOH to 11.0 L.
A) 0.28 M
B) 0.91 M
C) 1.1 M
D) 3.1 M
E) 3.9 M
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molality of a solution prepared by dissolving 86.9 g of diethyl ether,C4H10O,in 425 g of benzene,C6H6?
A) 0.362 m
B) 0.498 m
C) 2.01 m
D) 2.76 m
E) None of these choices is correct.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Raoult's Law relates the vapor pressure of a solvent above a solution to the mole fraction of the solvent and the vapor pressure of the pure solvent.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following aqueous solutions will have the lowest osmotic pressure?
A) 0.10 m KOH
B) 0.10 m RbCl
C) 0.05 m CaSO4
D) 0.05 m BaCl2
E) 0.10 m K2SO4
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
A 7.112 M solution of sulfuric acid (H2SO4)in water has a density of 1.395 g/mL.What is its molality?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Cinnamaldehyde ( 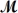 = 132.15 g/mol) is used as a flavoring agent.What mass of cinnamaldehyde must be added to 175 g of ethanol to give a solution whose boiling point is 82.7°C?
Kb = 1.22°C/m,boiling point of pure ethanol = 78.5°C
= 132.15 g/mol) is used as a flavoring agent.What mass of cinnamaldehyde must be added to 175 g of ethanol to give a solution whose boiling point is 82.7°C?
Kb = 1.22°C/m,boiling point of pure ethanol = 78.5°C
A) 62.4 g
B) 67.8 g
C) 76.2 g
D) 78.5 g
E) 79.6 g
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Two aqueous solutions are prepared: 1.00 m Na2CO3 and 1.00 m LiCl.Which of the following statements is true?
A) The Na2CO3 solution has a higher osmotic pressure and higher vapor pressure than the LiCl solution.
B) The Na2CO3 solution has a higher osmotic pressure and higher boiling point than the LiCl solution.
C) The Na2CO3 solution has a lower osmotic pressure and lower vapor pressure than the LiCI solution.
D) The Na2CO3 solution has a lower osmotic pressure and higher boiling point than the LiCl solution.
E) None of these statements is true.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Potassium fluoride is used for frosting glass.Calculate the molarity of a solution prepared by dissolving 78.6 g of KF in enough water to produce 225 mL of solution.
A) 0.304 M
B) 0.349 M
C) 1.35 M
D) 3.29 M
E) 6.01 M
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
If the density of a solution is less than 1.0 g/mL,its molarity will be greater than its molality.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calcium nitrite is used as a corrosion inhibitor in lubricants.What is the molality of a solution prepared by dissolving 18.5 g of calcium nitrite in 83.5 g of distilled water?
A) 0.0342 m
B) 0.0855 m
C) 0.222 m
D) 0.444 m
E) 1.68 m
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Two aqueous solutions are prepared: 2.0 m Cu(NO3) 2 and 2.0 m NaBr.Which of the following statements is true?
A) The Cu(NO3) 2 solution has a higher vapor pressure and lower freezing point than the NaBr solution.
B) The Cu(NO3) 2 solution has a higher vapor pressure and higher freezing point than the NaBr solution.
C) The Cu(NO3) 2 solution has a lower vapor pressure and lower freezing point than the NaBr solution.
D) The Cu(NO3) 2 solution has a lower vapor pressure and higher freezing point than the NaBr solution.
E) None of these statements is true.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Ideal solutions do not conform to Raoult's Law.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Safrole is used as a topical antiseptic.Calculate the vapor pressure of a solution prepared by dissolving 0.75 mol of safrole in 950 g of ethanol ( 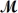 = 46.07 g/mol) .P°ethanol = 50.0 torr at 25°C.
= 46.07 g/mol) .P°ethanol = 50.0 torr at 25°C.
A) 1.8 torr
B) 11 torr
C) 15 torr
D) 40 torr
E) 48 torr
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many moles of solute particles are present in 100.0 mL of 2.50 M (NH4) 3PO4?
A) 0.100 mol
B) 0.250 mol
C) 0.500 mol
D) 0.750 mol
E) l.00 mol
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
If the shell of a raw egg is carefully dissolved away,and the egg in its flexible membrane is then placed in distilled water,the egg's volume will expand.Explain.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Human blood has a molar concentration of solutes of 0.30 M.What is the osmotic pressure of blood at 25°C?
A) 0.012 atm
B) 0.62 atm
C) 6.8 atm
D) 7.3 atm
E) >10.atm
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 73
Related Exams