Correct Answer

verified
a. The three structures and th...View Answer
Show Answer
Correct Answer
verified
View Answer
Essay
Draw Lewis structures which obey the octet rule, for the following atoms, molecules, and ions, showing all valence electrons. Central atoms are shown in bold. a. NH3 b. O3 (Hint: O3 is not cyclic) c. HCN d. SO3
Correct Answer

verified
A)11ec6d41_27c4_e72e_9cd1_85b54184eab2_T...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
In which one of the following molecules are all the bonds single?
A) O3
B) POCl3
C) CO
D) COCl2
E) N2H4
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the following Lewis structure for ClO3F, chlorine has a formal charge of ____ and an oxidation number of ____. 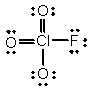
A) 7, 7
B) 7, -1
C) 1, 1
D) 1, -1
E) 1, 7
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct Lewis structure for NOCl, a reactive material used as an ionizing solvent.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) None of the above is correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use VSEPR theory to predict the electron group arrangement around iodine, the central atom in the ion IF2-.
A) octahedral
B) trigonal bipyramidal
C) tetrahedral
D) trigonal planar
E) bent
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the ideal bond angles in FNO using the molecular shape given by the VSEPR theory.
A) 90°
B) 109°
C) 120°
D) 180°
E) between 120 and 180°
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Oxygen difluoride is a powerful oxidizing and fluorinating agent. Select its Lewis structure.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) None of the above is correct.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of XeO2F2 as predicted by the VSEPR theory? 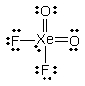
A) square planar
B) tetrahedral
C) square pyramidal
D) see-saw
E) octahedral
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the smallest actual bond angle in BrF3 using the VSEPR theory.
A) more than 120°
B) exactly 120°
C) between 109° and 120°
D) between 90° and 109°
E) less than 90°
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
The molecule AX2, where A and X are different elements, will have a dipole moment if the molecule is bent.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the ideal bond angles around nitrogen in N2F2 using the molecular shape given by the VSEPR theory. (The two N atoms are the central atoms.)
A) 90°
B) 109°
C) 120°
D) 180°
E) between 120 and 180°
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The formal charge on Cl in the structure shown for the perchlorate ion is 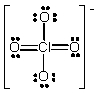
A) -2
B) -1
C) 0
D) +1
E) +2
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct Lewis structure for TeBr2.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the Lewis structure for XeO2F2 which correctly minimizes formal charges.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of BCl3 as predicted by the VSEPR theory?
A) linear
B) trigonal planar
C) bent
D) tetrahedral
E) trigonal pyramidal
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Essay
Using SO2 as an example, describe the sort of experimental data which might suggest that no single Lewis structure is an accurate representation of its bonding.
Correct Answer

verified
In the SO2 molecule, the two sulfur-oxyge...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which one of the following molecules contains a double bond?
A) N2
B) PCl5
C) CH2O
D) C2H2
E) I2
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following Lewis structures is definitely incorrect? 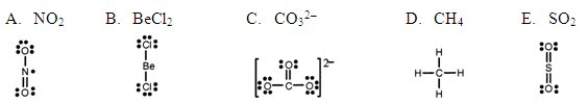
A) A
B) B
C) C
D) D
E) E
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of ClF2- as predicted by the VSEPR theory?
A) linear
B) bent
C) see-saw
D) T-shaped
E) L-shaped
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 109
Related Exams