B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 0.100 m MgSO4 solution has a freezing point of -0.23°C. What is the van't Hoff factor for this solution? Kf = 1.86°C/m
A) 0.62
B) 1.0
C) 1.2
D) 2.0
E) 4.0
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
In general, water is a good solvent for both polar and non-polar compounds.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Procaine hydrochloride ( 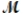 = 272.77 g/mol) is used as a local anesthetic. Calculate the molarity of a 4.666 m solution which has a density of 1.1066 g/mL.
= 272.77 g/mol) is used as a local anesthetic. Calculate the molarity of a 4.666 m solution which has a density of 1.1066 g/mL.
A) 2.272 M
B) 4.056 M
C) 4.216 M
D) 4.666 M
E) none of the above
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the type of interaction which best describes the attraction between Mg2+ ions and water molecules.
A) dipole-dipole
B) dipole-induced dipole
C) ion-dipole
D) ion-induced dipole
E) ion-hydrogen bond
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the strongest electrolyte from the following set.
A) CH3CH2OH, ethanol
B) LiNO3
C) C6H12O6, glucose
D) CCl4
E) HF
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An emulsion is a dispersion consisting of a
A) solid in a liquid.
B) liquid in a liquid.
C) gas in a liquid.
D) liquid in a solid.
E) gas in a solid.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the vapor pressure of a solution prepared by dissolving 0.500 mol of a non-volatile solute in 275 g of hexane ( 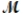 = 86.18 g/mol) at 49.6°C. P°hexane = 400.0 torr at 49.6°C.
= 86.18 g/mol) at 49.6°C. P°hexane = 400.0 torr at 49.6°C.
A) 54 torr
B) 154 torr
C) 246 torr
D) 346 torr
E) 400. torr
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Carbon tetrachloride, once widely used in fire extinguishers and as a dry cleaning fluid, has been found to cause liver damage to those exposed to its vapors over long periods of time. What is the boiling point of a solution prepared by dissolving 375 g of sulfur (S8, 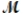 = 256.5 g/mol) in 1250 g of CCl4? Kb = 5.05°C/m, boiling point of pure CCl4 = 76.7°C?
= 256.5 g/mol) in 1250 g of CCl4? Kb = 5.05°C/m, boiling point of pure CCl4 = 76.7°C?
A) 70.8°C
B) 75.2°C
C) 78.2°C
D) 82.6°C
E) >85°C
G) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
Electrolyte solutions generally behave less ideally as the solute concentration increases.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
From the following list of aqueous solutions and water, select the one with the lowest freezing point.
A) 0.75 m (NH4) 3PO4
B) 1.0 m CaSO4
C) 1.0 m LiClO4
D) 1.5 m CH3OH, methyl alcohol
E) pure water
G) A) and E)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
A solution of ethanol (C2H6O) in water is sometimes used as a disinfectant. 1.00 L of this solution contains 553 g of ethanol and 335 g of water. What is the molality of the ethanol in this solution?
A) 0.392 m
B) 0.608 m
C) 13.5 m
D) 33.6 m
E) 35.8 m
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The most abundant molecules in the cell membranes of most species are
A) proteins.
B) sugars.
C) phospholipids.
D) fatty acids.
E) steroids.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 0.89% (w/v) sodium chloride solution is referred to as physiological saline solution because it has the same concentration of salts as human blood. What is the molarity of a physiological saline solution?
A) 0.0028 M
B) 0.015 M
C) 0.15 M
D) 0.30 M
E) 0.35 M
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Two aqueous are prepared: 1.00 m Na2CO3 and 1.00 m LiCl. Which of the following statements is true?
A) The Na2CO3 solution has a higher osmotic pressure and higher vapor pressure than the LiCl solution.
B) The Na2CO3 solution has a higher osmotic pressure and higher boiling point than the LiCl solution.
C) The Na2CO3 solution has a lower osmotic pressure and lower vapor pressure than the LiCI solution.
D) The Na2CO3 solution has a lower osmotic pressure and higher boiling point than the LiCl solution.
E) None of the above statements is true.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Cinnamaldehyde ( 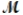 = 132.15 g/mol) is used as a flavoring agent. What mass of cinnamaldehyde must be added to 175 g of ethanol to give a solution whose boiling point is 82.7°C? Kb = 1.22°C/m, boiling point of pure ethanol = 78.5°C
= 132.15 g/mol) is used as a flavoring agent. What mass of cinnamaldehyde must be added to 175 g of ethanol to give a solution whose boiling point is 82.7°C? Kb = 1.22°C/m, boiling point of pure ethanol = 78.5°C
A) 62.4 g
B) 67.8 g
C) 76.2 g
D) 78.5 g
E) 79.6 g
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What concentration of aqueous FeCl3 would have the same osmotic pressure as a 0.20 M solution of CaCl2 at the same temperature, assuming ideal behavior?
A) 0.60 M
B) 0.40 M
C) 0.30 M
D) 0.15 M
E) 0.10 M
G) B) and E)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Saccharin, one of the first non-nutritive sweeteners used in soft-drinks, is 500 times sweeter than sugar in dilute aqueous solutions. The solubility of saccharin is 1.00 gram per 290 mL of solution. What is the molarity of a saturated saccharin solution? (  saccharin = 183.2 g/mol)
saccharin = 183.2 g/mol)
A) 0.0188 M
B) 0.632 M
C) 1.58 M
D) 3.45 M
E) none of the above
G) All of the above
Correct Answer

verified
A
Correct Answer
verified
True/False
Ideal solutions do not conform to Raoult's Law.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following pairs of ions is arranged so that the ion with the larger (i.e., more negative) heat of hydration is listed first?
A) Br-, K+
B) Mg2+, Sr 2+
C) Ca2+, Sc3+
D) Na+, Li+
E) None of the above are arranged in this way.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 105
Related Exams