A) 1 mole He(g) ; 1 mole Kr(g)
B) 1 mole O2(g) ; 2 mole O(g)
C) 1 mole CH4(g) ; 1 mole C2H6(g)
D) 1 mole Xe(g) at 1 atmosphere; 1 mole Xe(g) at 0.5 atmosphere
E) 20 one-dollar bills distributed randomly among 20 people; 20 one-dollar bills distributed randomly among 10 people
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following quantities used in thermodynamics: E, H, q, w, S, G. How many of them are state functions?
A) 0
B) 1
C) 2
D) 3
E) 4
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
For the reaction of xenon and fluorine gases to form solid XeF4, H° = -251 kJ and G° = -121 kJ at 25°C. Calculate S° for the reaction.
Correct Answer

verified
F...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
A reaction is proceeding toward equilibrium. At a certain stage, the concentrations of reactants and products are such that G = G°. What conclusion can reasonably be drawn about the reaction at this time?
A) K > Q
B) K < Q
C) K = Q
D) K = 1
E) Q = 1
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
The entropy of one mole of oxygen gas in a 0.5-L container is less than it would be in a 22.4-L container at the same temperature.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which relationship or statement best describes S° for the following reaction? 2H2S(g) + 3O2(g) 2H2O(g) + 2SO2(g)
A) .S° 0
B) .S° < 0
C) .S° > 0
D) .S° = H°/T
E) More information is needed to make a reasonable prediction.
G) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
The term microstate refers to the energy state of a single molecule in a system of many molecules.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When a sky diver free-falls through the air, the process is
A) non-spontaneous because he is accelerating due to the force applied by gravity.
B) non-spontaneous because he is losing potential energy.
C) non-spontaneous, if he had planned the jump for two weeks.
D) spontaneous.
E) in equilibrium.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
"A diamond is forever" is one of the most successful advertising slogans of all time. But is it true? For the reaction shown below, calculate the standard free energy change at 298K and determine whether or not a diamond is "forever". C(diamond) C(graphite) Data: Hf°(diamond) = 1.895 kJ/mol; S°(diamond) = 2.337 J mol-1 K-1; S°(graphite) = 5.740 J mol-1K-1.
A) .G° = 2.19 kJ; forever
B) .G° = -1.90 kJ; not forever
C) .G° = -2.90 kJ; not forever
D) .G° = 1.90 kJ; forever
E) .G° = < -1000 kJ; not forever
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following phase changes decreases the entropy of the system?
A) melting
B) heating a gas
C) vaporization
D) condensation
E) sublimation
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a process with S < 0, which one of the following statements is correct?
A) The process will definitely be spontaneous if H < 0.
B) The process will be definitely be spontaneous if H < TS.
C) The process can never be spontaneous.
D) The process will definitely be spontaneous, regardless of H.
E) The process will definitely be spontaneous if Ssurr > 0.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the equilibrium constant at 25°C for the reaction of methane with water to form carbon dioxide and hydrogen. The data refer to
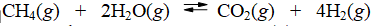
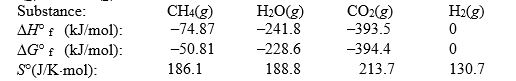
A) 8.2 × 1019
B) 0.96
C) 0.58
D) 1.2 × 10-20
E) 1.4 × 10-46
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following results in a decrease in the entropy of the system?
A) O2(g) , 300 K O2(g) , 400 K
B) H2O(s) , 0°C H2O(l) , 0°C
C) N2(g) , 25°C N2(aq) , 25°C
D) NH3(l) , -34.5°C NH3(g) , -34.5°C
E) 2H2O2(g) 2H2O(g) + O2(g)
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A certain process has Suniv > 0 at 25°C. What does one know about the process?
A) It is exothermic.
B) It is endothermic.
C) It is spontaneous at 25°C.
D) It will move rapidly toward equilibrium.
E) none of the above
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
The free energy of a perfect crystal at absolute zero, is zero.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which relationship or statement best describes S° for the following reaction? O3(g) + NO(g) O2(g) + NO2(g)
A) .S° 0
B) .S° < 0
C) .S° > 0
D) .S° = H°/T
E) More information is needed to make a reasonable prediction.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is always true for an exothermic process?
A) q sys > 0, Ssurr < 0
B) q sys < 0, Ssurr > 0
C) q sys < 0, Ssurr < 0
D) q sys > 0, Ssurr > 0
E) w < 0
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
In a spontaneous process, the entropy of the system always increases.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
In some spontaneous processes, the entropy of the surroundings decreases.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following pairs has the member with the greater molar entropy listed first? All systems are at 25°C.
A) CO(g) , CO2(g)
B) NaCl(s) , NaCl(aq)
C) H2S(g) , H2S(aq)
D) Li(s) , Pb(s)
E) H2(g) , H2O(g)
G) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 94
Related Exams