A) 0.0024 g
B) 0.078 g
C) 0.25 g
D) 0.50 g
E) none of the above
G) D) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The r-process occurs during supernova explosions.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the nuclide that completes the following nuclear reaction. 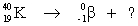
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following isotopes is definitely unstable?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The isotopes 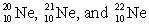 are all stable, while
are all stable, while 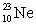 is radioactive. The mode of decay for
is radioactive. The mode of decay for 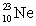 is most likely to be
is most likely to be
A) positron decay.
B) . decay.
C) . decay.
D) electron capture.
E) . decay.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Radioactive decay follows zero-order kinetics.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Positron decay and electron capture have the same net effect on the Z and N values of a nucleus.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following equations correctly represents electron capture by the 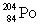 nucleus?
nucleus?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) D) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Gamma rays are not deflected by an electric field.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The isotopes of promethium, 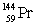 and
and 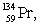 are unstable, and lie on opposite sides of the "line of stability". Which of the following combinations is most likely to represent the type of decay for these isotopes?
are unstable, and lie on opposite sides of the "line of stability". Which of the following combinations is most likely to represent the type of decay for these isotopes?
A) promethium-144, decay; promethium-134, positron decay
B) promethium-144, positron decay; promethium-134, decay
C) promethium-144, positron decay; promethium-134, electron capture
D) promethium-144, electron capture; promethium-134, positron decay
E) promethium-144, decay; promethium-134, decay
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Gamma rays are high energy electrons.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following nuclei has a magic number of neutrons and/or protons?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
After 4 half-lives, the fraction of a radioactive isotope which still remains is approximately one-eighth.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The s-process involves a slow succession of neutron absorption and beta decay processes during the normal life of a star.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The isotopes 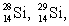 and
and 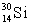 are all stable, while
are all stable, while 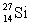 is radioactive. The mode of decay for
is radioactive. The mode of decay for 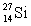 is most likely to be
is most likely to be
A) positron decay.
B) alpha decay.
C) beta decay.
D) gamma decay.
E) fission.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Fill in missing sub- and superscripts for all particles to complete the following equation for beta decay. 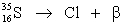
Correct Answer

verified
Correct Answer
verified
Essay
Explain how the number of protons and neutrons in a radioactive nucleus can be used to predict its probable mode of decay. Illustrate your answer with a schematic graph, properly labeled, showing stable nuclides (nuclei) in relation to number of protons and neutrons.
Correct Answer

verified
The "line of stability" on the accompany...View Answer
Show Answer
Correct Answer
verified
View Answer
Essay
Write a complete, balanced equation to represent the electron capture decay of argon-37.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 55-kg person exposed to thorium-234 receives 7.5 × 104 particles, each with an energy of 1.6 × 10-14 J. How many rads does the person receive?
A) 2.1 × 10-19
B) 1.2 × 10-17
C) 2.2 × 10-9
D) 1.2 × 10-9
E) none of the above
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
No alpha decay is observed for isotopes of elements with Z < 83.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 94
Related Exams