A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following peaks would you see in the IR spectrum of a carboxylic acid?
A) A flat line (carboxylic acids are not IR active)
B) A sharp line at 2250 cm-1
C) A broad peak from 3800-2800 cm-1
D) A broad peak from 800-600 cm-1
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How can you convert a carboxylic acid into an acid chloride?
A) Heat with hydrochloric acid
B) React with thionyl chloride (SOCl2)
C) React with sodium chloride
D) React with Cl2 and FeCl3
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which structure is consistent with the following 1H NMR spectrum? 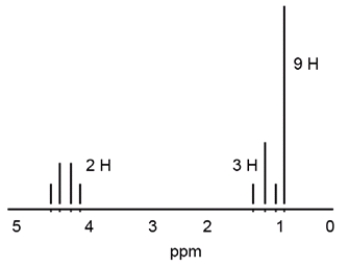
A) ethyl acetate
B) 2-propanone
C) 3,3-dimethyl-2-pentanone
D) ethyl 2,2-dimethylpropionate
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 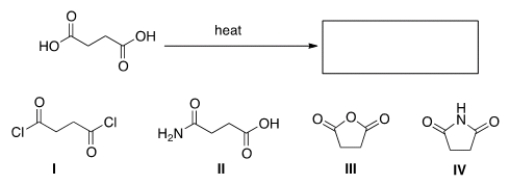
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An unknown compound has a sharp, medium peak at 2250 cm-1. The unknown compound is probably
A) an alkane
B) an aldehyde
C) an alkene
D) a nitrile
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name for the following compound? 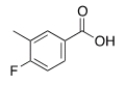
A) 4-fluoro-3-methylbenzoate
B) 4-fluoro-3-methylbenzoic acid
C) 3-methyl-4-fluorobenzoic acid
D) 4-fluoro-5-methylbenzoic acid
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Where is the carbonyl absorption found in the IR spectrum of a simple ester?
A) 2.5 ppm
B) 2250 cm-1
C) 3800 cm-1
D) 1740 cm-1
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a lactam? 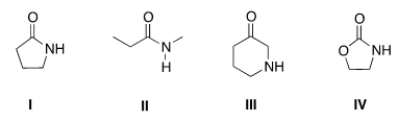
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How can you convert a carboxylic acid into an ester?
A) Heat with an alcohol and catalytic acid
B) Deprotonate with a base and react with an alcohol
C) Deprotonate with a base and react with an alkyl halide
D) Both (a) heat with an alcohol and catalytic acid and (c) deprotonate with a base and react with an alkyl halide
E) Both (a) heat with an alcohol and catalytic acid and (b) deprotonate with a base and react with an alcohol
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why doesn't nucleophilic acyl substitution stop at the tetrahedral intermediate?
A) The nucleophile is too basic.
B) Reforming the carbonyl is energetically favorable.
C) The leaving group is unstable and wants to be negatively charged.
D) There is no tetrahedral intermediate.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 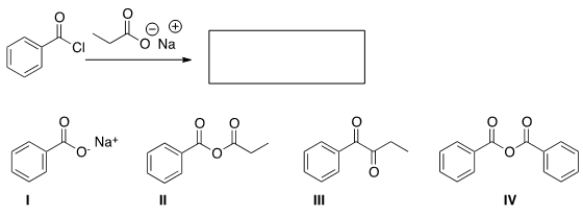
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 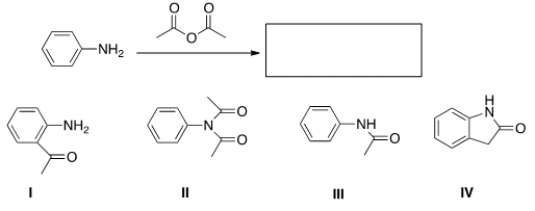
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What reagent would you use for the following transformation? 
A) LiAlH4
B) DIBAL-H
C) NaBH4
D) H2, Pd/C
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
You see an absorption at 2250 cm-1 in the IR spectrum of a compound. What kind of functional group is present?
A) A ketone
B) An aldehyde
C) An ester
D) A nitrile
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is an amide less reactive to nucleophilic acyl substitution than an acid chloride?
A) Nitrogen is a better leaving group.
B) Chloride is a better leaving group.
C) Nitrogen donates more electron density into the carbonyl.
D) The amide anion is less basic.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the structure for acetic propanoic anhydride? 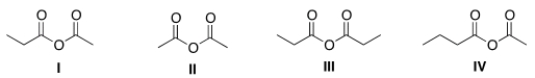
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the structure for the compound whose IUPAC name is pentyl 2-methylbutanoate? 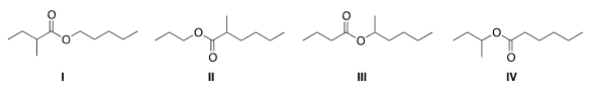
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name for the following compound? 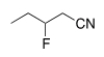
A) 2-fluorobutanonitrile
B) 2-fluoropentanonitrile
C) 3-fluoropentanonitrile
D) 2-fluorobutylcyanide
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Will the following reaction occur? 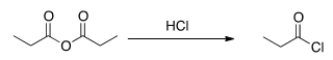
A) Yes
B) No
D) undefined
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 47
Related Exams