A) I > II > III
B) III > II > I
C) II > III > I
D) III > I > II
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the direction of equilibrium when acetylene (C2H2) reacts with ethoxide (CH3CH2O-) in an acid-base reaction? 
A) Left
B) Right
C) Neither
D) Cannot be determined
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the role of methylchloride (CH3Cl) in the following reaction? 
A) Lewis acid
B) Lewis base
C) Brønsted-Lowry acid
D) Brønsted-Lowry base
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a Lewis acid but not a Brønsted-Lowry acid?
A) CH3OH
B) H2O
C) CH3COOH
D) BF3
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following species is the conjugate base of the hydronium ion, H3O+?
A) H3O
B) H2O-
C) H2O
D) HO-
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following concepts can be used to explain the difference in acidity between acetylene (C2H2) and ethylene (C2H4) ?
A) Size
B) Resonance
C) Inductive effect
D) Hybridization
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of decreasing acidity, putting the most acidic first.
A) IV > II > III > I
B) III > II > IV > I
C) I > II > IV > III
D) III > IV > II > I
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following molecule with protons labeled, I-III. Rank these protons in order of decreasing acidity, putting the most acidic first. 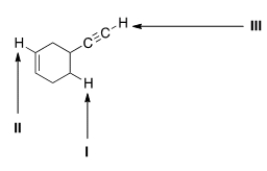
A) I > II > III
B) I > III > II
C) III > II > I
D) III > I > II
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is both a Brønsted-Lowry acid and base? I II III IV
A) I, II
B) I, III
C) II, IV
D) I, IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the nucleophilic site in the following compounds? 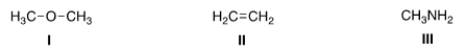
A) I = Hydrogen; II = electrons in bond; III = nitrogen.
B) I = Oxygen; II = carbon; III = nitrogen.
C) I = Hydrogen; II = carbon; III = carbon.
D) I = Oxygen; II = electrons in bond; III = nitrogen.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following concepts can be used to explain the difference in acidity between ethanol (CH3CH2OH) and 2-fluoroethanol (FCH2CH2OH) ?
A) Size
B) Inductive effect
C) Resonance
D) Hybridization
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ranks the compounds in order of increasing basicity, putting the least basic first?
A) CH3NH2 < CH3OH < CH4
B) CH3OH < CH3NH2 < CH4
C) CH4 < CH3NH2 < CH3OH
D) CH4 < CH3OH < CH3NH2
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the conjugate acid in the following reaction?
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is the weakest acid?
A) HF
B) HCl
C) HBr
D) HI
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the Lewis base in the following reaction. 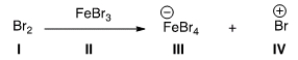
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of decreasing acidity, putting the most acidic first.
A) IV > II > III > I
B) IV > III > II > I
C) III > IV > II > I
D) III > IV > I > II
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the Lewis acid in the following reaction. 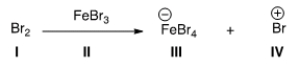
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about a Brønsted-Lowry base is true?
A) The net charge may be zero, positive, or negative.
B) All Brønsted-Lowry bases contain a lone pair of electrons or a bond.
C) All Brønsted-Lowry bases contain a proton.
D) The net charge may be zero or positive.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following species can be both Lewis acid and Lewis base? 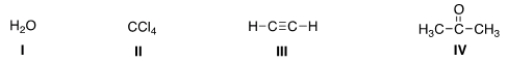
A) I, III, IV
B) I, II, IV
C) II, III, IV
D) I, II, III
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about Lewis bases is true?
A) Lewis bases are electron pair acceptors.
B) Lewis bases are electron pair donors.
C) Lewis bases are proton donors.
D) Lewis bases are proton acceptors.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 48
Related Exams