A) The product that is formed faster is the thermodynamic product.
B) The kinetic product predominates at low temperature.
C) The product that predominates at equilibrium is the kinetic product.
D) The thermodynamic product is less stable.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why would the compound below not react with a dienophile in a Diels-Alder reaction? 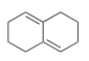
A) The compound is not a conjugated diene.
B) There are no electron withdrawing groups on the compound.
C) There are no electron donating groups on the compound.
D) The compound can not adopt the s-cis conformation.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the appropriate term for the mechanism of the addition of HBr to 1,3-dienes?
A) Nucleophilic addition
B) Electrophilic addition
C) Free radical addition
D) Conjugate addition
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How would you favor the formation of the kinetic product of the following reaction? 
A) Excess HBr
B) Long reaction time
C) Low temperature
D) High pressure
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 44 of 44
Related Exams