A) It is stronger.
B) It is weaker.
C) It has fewer hydrogens.
D) It makes a less stable cation.
F) A) and B)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Examine the IR below and classify the compound. 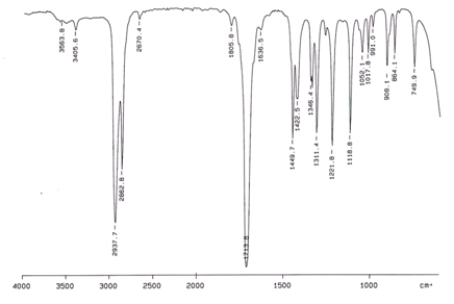
A) Alcohol
B) Aldehyde
C) Amine
D) Ketone
E) Carboxylic acid
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Stronger bonds will be found where in the infrared spectrum?
A) Higher molecular weight
B) Lower molecular weight
C) Lower wavenumbers
D) Higher wavenumbers
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Examine the IR below and classify the compound. 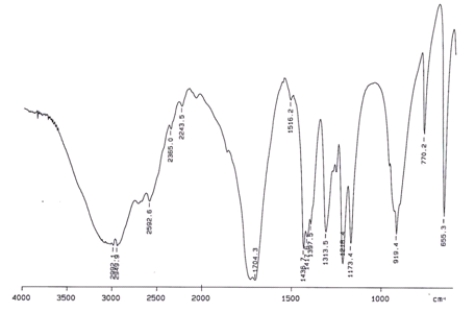
A) Alcohol
B) Aldehyde
C) Carboxylic acid
D) Ketone
F) All of the above
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
You are given a bottle of an organic liquid and told it must be either cyclohexane or 1-hexene. Which of the following statements is (are) true about these two compounds? 
A) The two compounds can be differentiated by their mass spectra because they will have molecular ion peaks at different m/z.
B) 1-Hexene will show an absorption at 1650 cm-1 but cyclohexane will not.
C) Both cyclohexane and 1-hexene will show C-H absorptions at about 2950 cm-1.
D) Statements B (1-Hexene will show an absorption at 1650 cm-1 but cyclohexane will not) and C (Both cyclohexane and 1-hexene will show C-H absorptions at about 2950 cm-1) are both true.
E) Statements A (The two compounds can be differentiated by their mass spectra because they will have molecular ions at different masses) , B (1-Hexene will show an absorption at 1650 cm-1 but cyclohexane will not) , and (Both cyclohexane and 1- hexene will show C-H absorptions at about 2950 cm-1) are all true.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following structures is consistent with a compound that displays a molecular ion peak at 56 and infrared signals at 2250 and 3600-3200 cm-1? 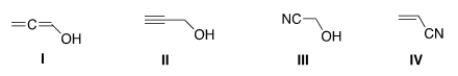
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is the infrared absorption for the stretching motion of internal alkynes rarely observed?
A) They do not form cations.
B) They are too strong.
C) Stretching in internal alkynes does not involve a change in dipole moment.
D) They do not have hydrogens.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following structures is consistent with a compound that displays a molecular ion peak at 103 and infrared signals at 2250 and 1600 cm-1? 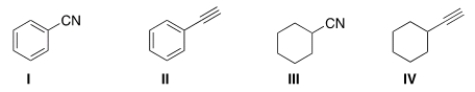
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What type of signal(s) would you observe in the mass and (or) infrared spectrum of the following compound? 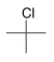
A) A signal at 1600 cm-1
B) A signal at 3300 cm-1
C) A single mass peak at 92 amu
D) Two mass peaks at 92 and 94 amu
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is (are) true about a compound that has a molecular ion peak in its mass spectrum at mass 104, and shows prominent peaks in its IR spectrum at 3200-2850 cm-1?
A) The compound has a molecular mass of 104.
B) The compound contains a C=O group and Csp3-H hybridized bonds.
C) The compound contains an OH group and Csp3-H hybridized bonds.
D) Both (The compound has a molecular mass of 104) and (The compound contains a C=O group and Csp3-H hybridized bonds) are true statements.
E) Both (The compound has a molecular mass of 104) and (The compound contains an OH group and Csp3-H hybridized bonds) are true statements.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is (are) accurate about the IR spectrum of compounds I, II, and III below? 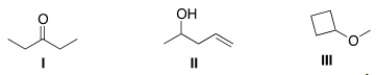
A) Compound I shows absorptions at 2950 and 1700 cm-1.
B) Compound II shows absorptions at 3200-3600 and 1650 cm-1.
C) Compound III shows absorptions at 3200-3600 and 2950 cm-1.
D) Both (Compound I shows absorptions at 2950 and 1700 cm-1) and (Compound II shows absorptions at 3200-3600 and 1650 cm-1) are true.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
You observe a compound that exhibits a mass spectrum with peak at 160 and a peak at 162, both of equal intensity. This compound contains:
A) Two chlorine atoms
B) One iodine atom
C) One bromine atom
D) Two bromine atoms
F) A) and B)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
A compound X has a molecular ion peak in its mass spectrum at m/z 136. What information does this tell us about X?
A) X has a molecular mass of 136.
B) The molecular formula for X is C8H8O2.
C) The empirical formula for X is C4H4O.
D) Both A (X has a molecular mass of 136) and B (The molecular formula for X is C8H8O2) are true.
E) Statements (X has a molecular mass of 136) , (The molecular formula for X is C8H8O2) , and (The empirical formula for X is C4H4O) are all true.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The functional group region of an infrared spectrum is:
A) Where the cations appear
B) Greater than or equal to 1500 cm-1
C) Less than 1500 cm-1
D) Greater than or equal to 2500 cm-1
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is (are) true about a compound that has molecular ion peaks in its mass spectrum at mass 170 and 172, and shows prominent peaks in its IR spectrum at 3150-3000 and 1600 cm-1?
A) The compound is not pure.
B) The compound contains a halogen.
C) The compound contains an OH group and Csp3-H hybridized bonds.
D) Both A (The compound is not pure) and B (The compound contains a halogen) are true statements.
E) Both A (The compound is not pure) and C (The compound contains an OH group and Csp3-H hybridized bonds) are true statements.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the three organic compounds drawn below. Which of the following statements is (are) true about the IR spectra of I, II, and III? 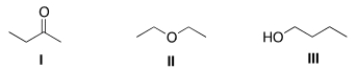
A) I shows strong absorptions at 2950 cm-1 and 1700 cm-1.
B) II shows strong absorptions at 2950 cm-1 and 2250 cm-1.
C) III shows strong absorptions at 2950 cm-1 and 3200-3600 cm-1.
D) Statements (I shows strong absorptions at 2950 cm-1 and 1700 cm-1) and (III shows strong absorptions at 2950 cm-1 and 3200-3600 cm-1) are true.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In an IR spectrum, which of the indicated C-H bonds exhibits a stretching absorption at the largest wave number? 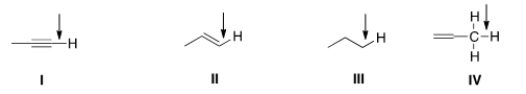
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The base peak in a mass spectrum corresponds to the most stable fragment. Propose a structure for a compound that is consistent with the following data.
(a) The molecular ion peak has m/z = 116
(b) The base peak is at m/z = 59.
(c) The compound is composed of C, H and O atoms.
(d) The IR spectrum shows a strong absorbance at 3257 cm-1. 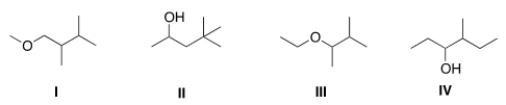
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following structures is consistent with a compound that displays a molecular ion peak at 84 and infrared signals at 3000-2850 cm-1 and no signals between 3000-3300 cm-1? 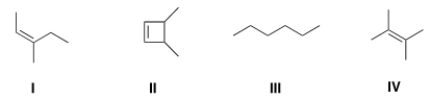
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following information is primarily obtained from an IR spectrum?
A) Conjugated system present in a compound.
B) Functional groups present in a compound.
C) Molecular weight of a compound.
D) The carbon and hydrogen framework of a compound.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 42
Related Exams