A) 1.32 atoms
B) 5.8 × 10-6 atoms
C) 2.4 × 1018 atoms
D) 3.3 × 1018 atoms
E) 3.5 × 1021 atoms
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the following reaction,identify X. 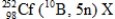 .
.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Alpha decay is not observed for isotopes of elements with atomic numbers less than 83.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many 14C atoms are in a charcoal sample that has a decay rate of 3500 disintegrations per min? (For 14C,t1/2 = 5730 yr.)
A) 2.9 × 107 atoms
B) 8.0 × 10-7 atoms
C) 1.4 × 1014 atoms
D) 1.5 × 1013 atoms
E) 6.02 × 1020 atoms
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
All isotopes of elements with atomic numbers higher than 83 (Bi)are radioactive.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the nuclear binding energy per nucleon for 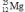 ? (1 kg = 6.022 × 1026 amu;
C = 2.99792458 × 108 m/s)
Particle Mass (amu)
? (1 kg = 6.022 × 1026 amu;
C = 2.99792458 × 108 m/s)
Particle Mass (amu) 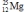 24.985839
24.985839 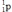 1.007276
1.007276 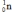 1.008665
1.008665
A) 0.214 J/nucleon
B) 3.20 × 10-11 J/nucleon
C) 1.28 × 10-12 J/nucleon
D) 0.999 J/nucleon
E) 7.35 × 10-11 J/nucleon
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the missing species in the following nuclear transmutation. 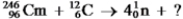 .
.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) None of the answers is correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the missing species in the following nuclear transmutation. 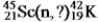
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the nuclear binding energy per nucleon of uranium-234? Particle Mass (amu) U-234 234) 040947 Neutron 1) 008701 Proton 1) 007316 Electron 0) 000549 [1 kg = 6.022 × 1026 amu; NA = 6.022 × 1023 mol-1; c = 2.99792458 × 108 m/s]
A) 2.75 × 10-10 J/nucleon
B) 3.04 × 10-10 J/nucleon
C) 1.38 × 10-12 J/nucleon
D) 1.27 × 10-12 J/nucleon
E) 1.18 × 10-12 J/nucleon
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A typical radius of an atomic nucleus is about
A) 100 μm.
B) 5000 mm.
C) 100 nm.
D) 5 × 10-3 pm.
E) 500 pm.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the nuclear binding energy per nucleon of potassium-40? Particle Mass (amu) K-40 39) 9632591 Neutron 1) 008701 Proton 1) 007316 Electron 0) 000549 (1 kg = 6.022 × 1026 amu; NA = 6.022 × 1023 mol-1; C = 2.99792458 × 108 m/s)
A) 1.33 × 10-12 J/nucleon
B) 5.33 × 10-11 J/nucleon
C) 5.64 × 10-11 J/nucleon
D) 1.41 × 10-12 J/nucleon
E) 2.97 × 10-12 J/nucleon
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which isotope,when bombarded with bismuth-209,would yield two neutrons and an isotope with atomic number 121 and mass number 299?
A) Pb-211
B) Po-209
C) Sr-92
D) Rn-38
E) Sr-38
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calcium-39 undergoes positron decay.Each positron carries 5.49 MeV of energy.How much energy will be emitted when 0.00250 mol of calcium-39 decays? (1 MeV = 1.602 × 10-13 J)
A) 13.2 kJ
B) 1.32 × 104 kJ
C) 1.32 × 106 kJ
D) 1.32 × 109 kJ
E) None of these choices is correct.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 30.0-kg child receives 2.65 × 107 β particles,each with an energy of 4.60 × 10-13 J.If the RBE = 0.78,how many millirem did the child receive?
A) 3.2 × 10-7 millirem
B) 5.2 × 10-7 millirem
C) 5.2 × 10-4 millirem
D) 3.2 × 10-2 millirem
E) None of these choices is correct.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How old is the oldest object that can reliably be dated using 14C dating?
A) 5730 years
B) 30,000 years
C) 60,000 years
D) 10,000 years
E) There is no limit to how old an object can be.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which equation correctly represents positron decay of 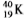
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When atoms of beryllium-9 are bombarded with alpha particles,neutrons are produced.What new isotope is also formed? 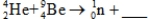 .
.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
Nuclear fission is the process in which a heavy nucleus (mass number > 200)divides to form smaller nuclei of intermediate mass and one or more protons.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A pure sample of tritium,3H,was prepared and sealed in a container for a number of years.Tritium undergoes β decay with a half-life of 12.32 years.How long has the container been sealed if analysis of the contents shows there are 5.25 mol of 3H and 6.35 mol of 3He present?
A) 2.34 yr
B) 3.38 yr
C) 9.77 yr
D) 14.1 yr
E) 25.6 yr
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
An alpha particle is a helium atom.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 125
Related Exams