A) 0.029 yr
B) 7.4 yr
C) 9.1 yr
D) 16 yr
E) 35 yr
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A mass of 6.02 × 1026 amu is equivalent to
A) 1 J.
B) 1 kg.
C) 1 g.
D) 1 mol.
E) 1 mg.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A sample of a radioisotope shows an activity of 999 disintegrations per minute due to beta decay.If after 1.10 years the activity is 952 disintegrations per minute,what is the half-life of this radioisotope?
A) 4.38 × 10-2 yr
B) 11.4 yr
C) 0.25 yr
D) 15.8 yr
E) 9.1 yr
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Nuclear fusion is the combination of small nuclei into larger ones.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the equation below,what particle or type of radiation needs to be included to balance the equation? 208Po → ? + 208At
A) Gamma particle
B) Alpha particle
C) Proton
D) Beta particle
E) Positron
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement is false?
A) Fission occurs among the heaviest isotopes,whereas fusion occurs more readily for light isotopes.
B) The mass defect (Δm) for a fission reaction is negative,whereas Δm for fusion is positive.
C) In order for fusion reactions to occur,temperatures must be in the millions of degrees.
D) The fission of Pu-239 atoms produces isotopes of many different elements.
E) Neutron-induced fission processes can occur at room temperature,rather than at millions of degrees.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Charcoal found under a stone at Stonehenge,England,has a carbon-14 activity that is 0.60 that of new wood.How old is the charcoal? (The half-life of carbon-14 is 5730 years.)
A) Less than 5730 yr
B) Between 5730 and 11,460 yr
C) Between 11,460 and 17,190 yr
D) More than 17,190 yr
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The half-life of 14C is 5730 yr.Assuming some charcoal from a campfire 29,000 years old was found,what fraction of the original C-14 would remain today?
A) 3.0 × 10-2
B) 0.20
C) 3.5
D) 0.33
E) 0.29
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Cesium-134 is a β emitter with a half-life of 2.0 years.How much of a 2.50-g sample of cesium-134 will remain after 10 years?
A) 0.0024 g
B) 0.078 g
C) 0.25 g
D) 0.50 g
E) 80.0 g
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Beta particles are identical to
A) protons.
B) helium atoms.
C) hydrogen atoms.
D) helium nuclei.
E) electrons.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which equation correctly represents electron capture by the 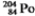 nucleus?
nucleus?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What element is the stable end-product of the uranium radioactive decay series?
A) Th
B) Pu
C) Ra
D) Au
E) Pb
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name for spontaneous emission of particles or electromagnetic radiation by certain nuclei?
A) Protons
B) Isotopes
C) Radioactivity
D) Neutrons
E) Electrons
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Iodine-131,t1/2 = 8.0 days,is used in the diagnosis and treatment of thyroid gland diseases.If a laboratory sample of iodine-131 initially emits 9.95 × 1018 β particles per day,how long will it take for the activity to drop to 6.22 × 1017 β particles per day?
A) 2.0 days
B) 16 days
C) 32 days
D) 128 days
E) None of the answers is correct.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the total binding energy of 12C? [12C atomic mass = 12.000 amu; mass of proton = 1.007825 amu; mass of neutron = 1.008665 amu; 1 kg =6.022 x 1026amu; 1 J = 1 (kg•m2) /s2]?
A) 1.80 x 10-9 J
B) 1.48 x 10-8 J
C) 3.60 x 10-9 J
D) 8.91 x 1015 J
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the missing symbol in this plutonium fission reaction? 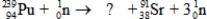
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the energy equivalent of 1 amu? (c = 2.99792458 × 108 m/s)
A) 5.0 × 10-19 J
B) 5.4 × 1043 J
C) 6.6 × 109 J
D) 1.5 × 10-10 J
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name for the difference between the mass of an atom and the sum of the masses of its constituent nucleons?
A) Gamma particle
B) Alpha particle
C) Mass difference
D) Beta particle
E) Mass defect
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Most nuclear reactors in the United States are __________.
A) heavy water reactors
B) fusion reactors
C) breeder reactors
D) tokamaks
E) light water reactors
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following isotopes is least likely to be stable?
A) (40Ca)
B) (32S)
C) (30Al)
D) (55Mn)
E) (119Sn)
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 125
Related Exams