B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The most basic oxides are formed from elements found in the __________________ region of the periodic table.
A) upper right
B) upper left
C) center
D) lower right
E) lower left
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 0.15 M solution of chloroacetic acid has a pH of 1.86.What is the value of Ka for this acid?
A) 7.2 × 101
B) 1.6 × 10-1
C) 9.9 × 10-2
D) 1.4 × 10-3
E) 2.7 × 10-4
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the conjugate base of NH3?
A) NH2
B) NH2+
C) NH2-
D) NH4
E) NH4+
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Picric acid has been used in the leather industry and in etching copper.However,its laboratory use has been restricted because it dehydrates on standing and can become shock sensitive.It has an acid dissociation constant of 0.42.What is the [H3O+] for a 0.20 M solution of picric acid?
A) 0.022 M
B) 0.052 M
C) 0.15 M
D) 0.20 M
E) 0.29 M
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the concentration of malonate ion (C3H2O42-) in a 0.200 M solution of malonic acid (C3H4O4) .(For malonic acid,Ka1 = 1.4 × 10-3,Ka2 = 2.0 × 10-6.)
A) 2.8 × 10-4 M
B) 0.016 M
C) 1.8 × 10-4 M
D) 1.4 × 10-3 M
E) 2.0 × 10-6 M
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The first ionization constant Ka1 is always smaller than the second ionization constant Ka2 for the ionization of a diprotic acid.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is a basic oxide?
A) CO2
B) MgO
C) As2O3
D) SO2
E) Cl2O7
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
Amphoteric oxides exhibit both acidic and basic properties.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the pH of a 0.021 M NaCN solution.[Ka(HCN) = 4.9 × 10-10]
A) 1.68
B) 3.19
C) 4.69
D) 9.31
E) 10.81
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is [OH-] for a solution at 25°C that has [H3O+] = 8.23 × 10-2 M?
A) 8.23 × 10-2 M
B) 1.22 × 10-6 M
C) 8.23 × 10-12 M
D) 1.22 × 10-13 M
E) 8.23 × 10-16 M
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is an acidic oxide?
A) P4O10
B) MgO
C) Fe2O3
D) K2O
E) Cr2O3
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Below is a representation of pure liquid HX,an amphoteric substance,at equilibrium.(Each circle represents 1.0 ×10-3 mol of atoms,and the volume of the box is 1.0 L.) 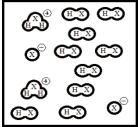 What is the autoionization constant for HX?
What is the autoionization constant for HX?
A) 4.9 ×10-2
B) 4.0 ×10-3
C) 4.4 ×10-4
D) 4.0 ×10-6
E) 2.5 ×105
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Acid strength decreases in the series HI > HSO4-> HF > HCN.Which of these anions is the weakest base?
A) I-
B) SO42-
C) F-
D) CN-
F) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
The following is the correct order for the acid strength for these oxoacids.HClO > HClO2 > HClO3> HClO4
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Equal volumes of two solutions,one containing a strong acid at pH 2 and the other containing a strong base at pH 12,are mixed.Once equilibrium is achieved,what is the final pH of the combined solution?
A) 6
B) 7
C) 10
D) 14
E) 24
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the correct Kb expression for the reaction below? B(aq) + H2O (l) ![Which of the following is the correct Kb expression for the reaction below? B(aq) + H<sub>2</sub>O (l) HB+(aq) + OH<sup>-</sup>(aq) A) K<sub>b</sub> = [B][ H<sub>2</sub>O] B) C) D) E) K<sub>b</sub> = [HB<sup>+</sup>][OH<sup>-</sup>]](https://d2lvgg3v3hfg70.cloudfront.net/TB6485/11eab46b_9ca1_7ad2_a889_7bb112cd6732_TB6485_11.jpg) HB+(aq) + OH-(aq)
HB+(aq) + OH-(aq)
A) Kb = [B][ H2O]
B) ![]()
C) ![]()
D) ![]()
E) Kb = [HB+][OH-]
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Since zirconium is a metal,ZrO2 is expected to be a/an _____ oxide.
A) acidic
B) ionic
C) amphoteric
D) neutral
E) basic
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The ammonium ion,NH4+,is a weak acid.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is a Lewis acid but not a Brønsted acid?
A) HCN
B) CO32-
C) OH-
D) Cl-
E) Al3+
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 133
Related Exams