A) 0.00700 M
B) 0.0164 M
C) 0.0383 M
D) 0.0230 M
E) 0.0575 M
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the oxidizing agent in the chemical reaction 2MnO4- + 5H2SO3→ 2Mn2+ + 5SO42- + 4H+ + 3H2O.
A) MnO4-
B) H2SO3
C) Mn2+
D) SO42-
E) H+
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Based on the solubility rules,which one of these compounds is soluble in water?
A) Hg2Cl2
B) Na2S
C) Ag2CO3
D) Ag2S
E) BaCO3
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the reaction depicted below,if Z represents Zn,which metal could A represent? 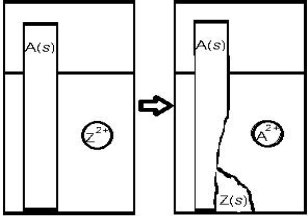
A) Al
B) Na
C) Pb
D) Ca
E) Cu
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is present in all aqueous Brønsted acid solutions?
A) H2
B) H+
C) OH-
D) H2SO4
E) Cl-
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Complete the following reaction and identify the Brønsted base: NaOH(aq) + HCl(aq) →
A) Na(aq) + HOCl(aq) ; NaOH is the base.
B) NaCl(aq) + OH-(aq) ; HCl is the base.
C) NaCl(aq) + OH-(aq) ; NaOH is the base.
D) H2O(l) + NaCl(aq) ; HCl is the base.
E) NaCl(aq) + H2O(l) ; NaOH is the base.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If aqueous solutions of KMnO4,FeSO4 and H2SO4,are mixed,which ions are spectator ions? 2KMnO4(aq) + 10FeSO4(aq) + 8H2SO4(aq) → K2SO4(aq) + 2MnSO4(aq) + 5Fe2(SO4) 3(aq) + 8H2O(l)
A) only K+
B) only SO42-
C) only K+ and SO42-
D) only K+,SO42- ,and Fe2+
E) only K+,SO42-,Fe2+,and Mn2+
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calcium nitrate tetrahydrate dissolves in warm water to the extent of 266 g per 100.cm3.What is the concentration of nitrate ions in this solution?
A) 32.4 M
B) 22.5 M
C) 11.3 M
D) 16.2 M
E) 8.10 M
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The substance NH3 is considered to be
A) a weak acid.
B) a weak base.
C) a strong acid.
D) a strong base.
E) neither acidic nor basic.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The distinguishing characteristic of all nonelectrolyte solutions is that they
A) contain ions.
B) do not conduct electricity.
C) react with other solutions.
D) always contain acids.
E) conducts heat.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the oxidation number of N in N2H4?
A) +4
B) +2
C) -2
D) -4
E) 0
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the major ions present in an aqueous HNO3 solution.
A) HN2+,O2-
B) OH-,NO3-
C) OH-,NO+
D) H+,N3-,O2-
E) H+,NO3-
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Based on the solubility rules,which one of these compounds is soluble in water?
A) AgBr
B) AgCl
C) Ag2CO3
D) AgNO3
E) Ag2S
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Suppose aqueous solutions of silver(I) nitrate and potassium chloride are mixed.Which represents the likely result?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 50.0 mL sample of 0.436 M NH4NO3 is diluted with water to a total volume of 250.0 mL.What is the ammonium nitrate concentration in the resulting solution?
A) 2.18 M
B) 0.523 M
C) 0.349 M
D) 0.174 M
E) 0.0872 M
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is oxidized in the following reaction? Fe + Ag2O → FeO + 2Ag
A) Ag
B) Ag2O
C) Fe
D) FeO
E) 2Ag
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is soluble in water?
A) AgCl
B) Rb2CrO4
C) Hg2I2
D) CaSO4
E) PbBr2
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the net ionic equation for the reaction between aqueous solutions of lithium hydroxide and hydrobromic acid? LiOH(aq) + HBr(aq) → H2O(l) + LiBr(aq)
A) LiOH(aq) → Li+(aq) + OH-(aq)
B) HBr(aq) → H+(aq) + Br-(aq)
C) H+(aq) + OH-(aq) → H2O(l)
D) Li+(aq) + Br-(aq) → LiBr(aq)
E) Li+(aq) + OH-(aq) + H+(aq) + Br-(aq) → H2O(l) + LiBr(aq)
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the reducing agent in the following reaction? Mg + NiO2 + 2H2O → Mg(OH) 2 + Ni(OH) 2
A) Mg
B) NiO2
C) H2O
D) Mg(OH) 2
E) Ni(OH) 2
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many moles of H+(aq) ions are present in 750 mL of 0.65 M hydrochloric acid?
A) 1.2 mol
B) 0.98 mol
C) 0.87 mol
D) 0.65 mol
E) 0.49 mol
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 144
Related Exams